- Remote Access
- Save figures into PowerPoint
- Download tables as PDFs


34: Tuberculosis
David Cluck
- Download Chapter PDF
Disclaimer: These citations have been automatically generated based on the information we have and it may not be 100% accurate. Please consult the latest official manual style if you have any questions regarding the format accuracy.
Download citation file:
- Search Book
Jump to a Section
Patient presentation.
- Full Chapter
- Supplementary Content
Chief Complaint
“I have a cough that won’t go away.”
History of Present Illness
A 63-year-old male presents to the emergency department with complaints of cough/shortness of breath which he attributes to a “nagging cold.” He states he fears this may be something worse after experiencing hemoptysis for the past 3 days. He also admits to waking up in the middle of the night “drenched in sweat” for the past few weeks. When asked, the patient denies ever having a positive PPD and was last screened “several years ago.” His chart indicates he was in the emergency department last week with similar symptoms and was diagnosed with community-acquired pneumonia and discharged with azithromycin.
Past Medical History
Hypertension, dyslipidemia, COPD, atrial fibrillation, generalized anxiety disorder
Surgical History
Appendectomy at age 18
Family History
Father passed away from a myocardial infarction 4 years ago; mother had type 2 DM and passed away from a ruptured abdominal aortic aneurysm
Social History
Retired geologist recently moved from India to live with his son who is currently in medical school in upstate New York. Smoked ½ ppd × 40 years and drinks 6 to 8 beers per day, recently admits to drinking ½ pint of vodka “every few days” since the passing of his wife 6 months ago.
Sulfa (hives); penicillin (nausea/vomiting); shellfish (itching)
Home Medications
Albuterol metered-dose-inhaler 2 puffs q4h PRN shortness of breath
Aspirin 81 mg PO daily
Atorvastatin 40 mg PO daily
Budesonide/formoterol 160 mcg/4.5 mcg 2 inhalations BID
Clonazepam 0.5 mg PO three times daily PRN anxiety
Lisinopril 20 mg PO daily
Metoprolol succinate 100 mg PO daily
Tiotropium 2 inhalations once daily
Venlafaxine 150 mg PO daily
Warfarin 7.5 mg PO daily
Physical Examination
Vital signs.
Temp 100.8°F, P 96, RR 24 breaths per minute, BP 150/84 mm Hg, pO 2 92%, Ht 5′10″, Wt 56.4 kg
Slightly disheveled male in mild-to-moderate distress
Normocephalic, atraumatic, PERRLA, EOMI, pale/dry mucous membranes and conjunctiva, poor dentition
Bronchial breath sounds in RUL
Cardiovascular
NSR, no m/r/g
Soft, non-distended, non-tender, (+) bowel sounds
Genitourinary
Get free access through your institution, pop-up div successfully displayed.
This div only appears when the trigger link is hovered over. Otherwise it is hidden from view.
Please Wait
CASE STUDY ON PULMONARY TUBERCULOSIS
- January 2017
- This person is not on ResearchGate, or hasn't claimed this research yet.

- Tamil Nadu Dr. M.G.R. Medical University

- Vel's Group of Institutions
Discover the world's research
- 25+ million members
- 160+ million publication pages
- 2.3+ billion citations

- J T Crawford
- T M Shinnick
- JL Kamerbeek

- EMERG INFECT DIS
- Gaby E. Pfyffer

- Dick van Soolingen
- Mamadou Daffé
- Michael R. McNeil
- Patrick J. Brennan
- D Soolingen
- T Hoogenboezem
- Pwm Hermans
- K S Teppema
- Recruit researchers
- Join for free
- Login Email Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google Welcome back! Please log in. Email · Hint Tip: Most researchers use their institutional email address as their ResearchGate login Password Forgot password? Keep me logged in Log in or Continue with Google No account? Sign up
- Case report
- Open access
- Published: 19 November 2022
A case report of persistent drug-sensitive pulmonary tuberculosis after treatment completion
- Sergo A. Vashakidze 1 , 2 ,
- Abivarma Chandrakumaran 3 ,
- Merab Japaridze 1 ,
- Giorgi Gogishvili 1 ,
- Jeffrey M. Collins 4 ,
- Manana Rekhviashvili 1 &
- Russell R. Kempker 4
BMC Infectious Diseases volume 22 , Article number: 864 ( 2022 ) Cite this article
6644 Accesses
1 Citations
2 Altmetric
Metrics details
Mycobacterium tuberculosis (Mtb) has been found to persist within cavities in patients who have completed their anti-tuberculosis therapy. The clinical implications of Mtb persistence after therapy include recurrence of disease and destructive changes within the lungs. Data on residual changes in patients who completed anti-tuberculosis therapy are scarce. This case highlights the radiological and pathological changes that persist after anti-tuberculosis therapy completion and the importance of achieving sterilization of cavities in order to prevent these changes.
Case presentation
This is a case report of a 33 year old female with drug-sensitive pulmonary tuberculosis who despite successfully completing standard 6-month treatment had persistent changes in her lungs on radiological imaging. The patient underwent multiple adjunctive surgeries to resect cavitary lesions, which were culture positive for Mtb. After surgical treatment, the patient’s chest radiographies improved, symptoms subsided, and she was given a definition of cure.
Conclusions
Medical therapy alone, in the presence of severe cavitary lung lesions may not be able to achieve sterilizing cure in all cases. Cavities can not only cause reactivation but also drive inflammatory changes and subsequent lung damage leading to airflow obstruction, bronchiectasis, and fibrosis. Surgical removal of these foci of bacilli can be an effective adjunctive treatment necessary for a sterilizing cure and improved long term lung health.
Peer Review reports
Mycobacterium tuberculosis treatment has been evolving over the years, especially with the introduction of newer drugs and shorter regimens [ 1 , 2 ]. Apart from the cavitary nature of tuberculous disease, patients who have been treated with current regimens often are given the designation of cure without achieving proper sterilization. Patients who complete the tuberculous regimen are given the definition of cure after they achieve sputum negativity but many of these patients harbor bacilli within cavities that continue to exert their effects on the respiratory system [ 3 ]. The residual changes that occur in patients who have completed medical therapy have been poorly attended to in the literature. Patients that underwent surgical and medical sterilization have been reported to have better pulmonary health in the long term, especially after the removal of cavities [ 4 ].
Here, we report a patient who underwent a complete regimen of medical therapy for pulmonary tuberculosis and later had to have surgical resection of her cavities, which grew tuberculous bacilli even after achieving sputum negativity.
A 33-year-old female from the country of Georgia presented to a tuberculosis dispensary on July 10, 2020, with a temperature of 38° C and symptoms of malaise, productive cough, and night sweats. The patient had no known medical problems. She reported smoking ~ 10 cigarettes daily and denied alcohol or illicit drug use. She had 3 children and her husband was a prisoner being treated for pulmonary tuberculosis. Upon physical examination there were decreased breath sounds in the upper lobes of the lungs with dullness to percussion. The patient had a body mass index (BMI) of 16.3 kg/m 2 . A complete blood count revealed a moderate leukocytosis of 10.2 × 10 9 /L and an erythrocyte sedimentation rate (ESR) of 42 mm/h. Biochemical blood parameters were normal. Sputum testing found a negative acid-fast bacilli (AFB) microscopy, positive Xpert MTB/RIF test (no RIF resistance), and positive culture for Mycobacterium tuberculosis (Mtb). Additionally, drug susceptibility testing (DST) revealed sensitivity to rifampin, isoniazid, and ethambutol. Chest radiography revealed multiple small foci in the upper lobes of both lungs and a cavity in the right lung (Fig. 1 A). The patient was initiated on daily outpatient treatment with three pills of a fixed dosed combination pill containing isoniazid 75 mg, rifampin 150 mg, ethambutol 275 mg and pyrazinamide 400 mg. Treatment was given through directly observed therapy (DOT). She converted her sputum cultures to negative at 2 months and continued rifampin and isoniazid to finish 6 months of treatment. An end of treatment chest x-ray revealed fibrosis and honeycombing in the right upper lung, and fibrosis and dense focal shadows in the 1st and 2nd intercostal spaces of the left lung (Fig. 1 B). The complete treatment timeline is summarized in Fig. 2 .
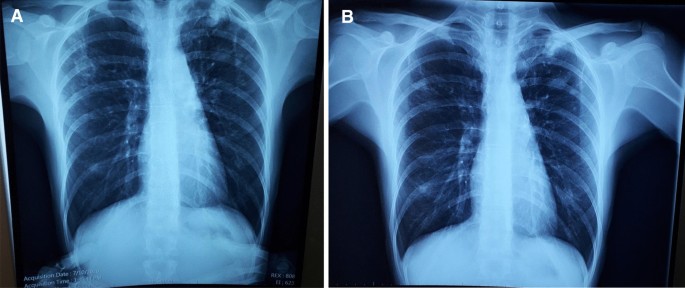
A (left): Baseline chest X-ray showing a cavity in the right lung and multiple foci in the upper lobes of both lungs. B (right): End of initial treatment chest X-ray, showing fibrosis, local honeycombing and dense focal shadows in both lungs
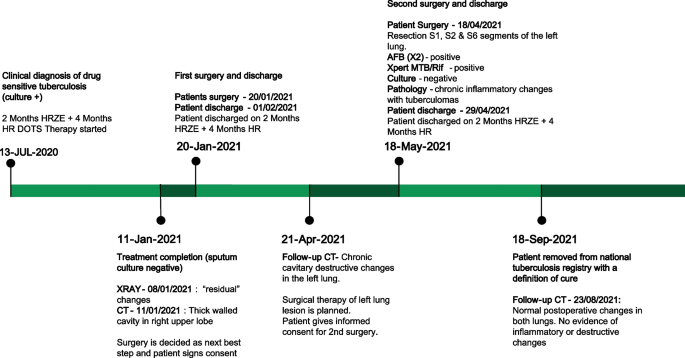
Patient treatment timeline ( HRZE isoniazid, rifampin, pyrazinamide, ethambutol; HR isoniazid & rifampin; DOTS directly observed therapy, short-course; CT computed tomography; AFB acid fast bacilli)
A follow up chest computed tomography (CT) scan demonstrated a cavity in the right upper lobe measuring 12 × 10 mm in size with a thick and heterogeneous wall and nodules and bronchiectasis in the left lung (Fig. 3 A–D). Based on CT findings and in accordance with National tuberculosis guidelines, the patient was offered surgical resection of the affected portion of the lung. It should be noted that the patient reported no symptoms, complaints, or functional disability before the surgery. Preoperative workup including pulmonary function testing, an echocardiogram, bronchoscopy, and blood chemistries were normal. The patient consented to surgery and underwent a surgical resection of the S1 and S2 segments of the right lung 2 weeks later. Intraoperatively, moderate adhesions were visualized in the S1 and S2 area with a palpable dense formation ~ 3.0 cm in diameter, in addition to a dense nodule. Gross pathology of the resected lesion showed a thick-walled fibrous cavity filled with caseous necrosis (Fig. 4 A) corresponding to the right preoperative CT lesion seen on Fig. 3 A, C.
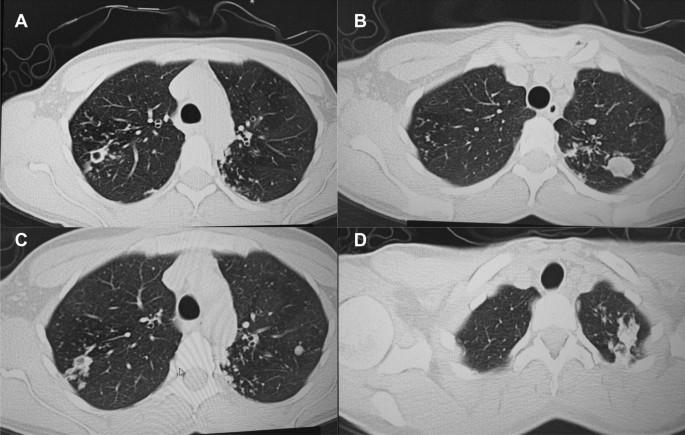
CT scan (January 11, 2021) showing, A a cavity in the upper lobe of the right lung with heterogeneous thick walls. B S1 and S2 segments of the left lung shows a 23 × 18 mm oval shaped calcified inclusions; C , D areas with calcified, compacted nodules 13 × 20 mm in size with additional traction bronchiectasis
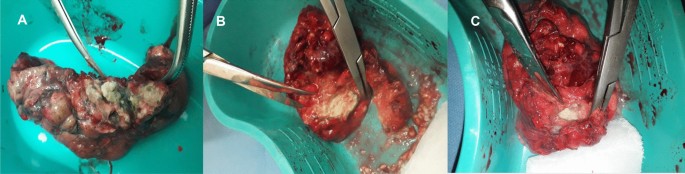
A Gross pathological image of a resected cavity with caseous material from first surgery (S1 & S2 segment of right lung). B The gross pathology from the second surgery showed the presence of a blocked cavity measuring up to 2 cm in diameter filled with caseous material in the S1, S2 and C Tuberculoma in S6 segment
Microbiological analysis on the resected tissue revealed acid-fast bacilli on microscopy, and positive Xpert MTB/RIF and culture results. Mtb grew from the caseous center, inner and outer walls of the cavity and a resected foci located ~ 3 cm from the cavity. DST revealed sensitivity to isoniazid, rifampin, and ethambutol.
Pathological examination of the resected lesion showed findings consistent with fibrocavernous tuberculosis. No postoperative complications were experienced, and the patient reinitiated first-line therapy via DOT on the 2nd postoperative day and was discharged on postoperative day 11.
A follow up CT scan performed after 3 months showed postoperative changes in the right upper lobe, and an unchanged left lung (Fig. 5 A–C). Based on the persistent conglomerate of tuberculomas and multiple small tuberculous foci, growth of Mtb from the previous surgical specimen, and the patient’s social situation (mother of three young children) a second surgery to optimize the chance of cure was recommended. The patient reported no symptoms, complaints, or functional disability before the surgery. Preoperative sputum testing found negative AFB smear microscopy and culture. The patient underwent the second operation on May 18, 2021, in which the S1, S2 and part of the S6 segment of the left lung were resected. Intraoperatively, moderate adhesions seen along with a dense palpable ~ 3 cm mass in the S1 and S2 region and a dense focus in S6.
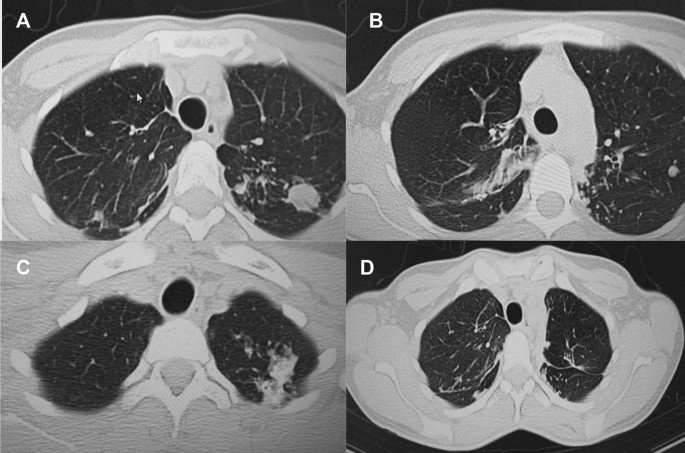
A – C Follow-up CT scan after first adjunctive surgery showing postoperative changes of the right lung and radiological changes in the left lung, that were unchanged compared to the initial CT. D Final CT scan showing normal postoperative changes with no cavities as previously seen
Microbiological examinations performed on resected tissue revealed positive AFB smear microscopy and Xpert MTB/RIF results and a negative AFB culture. The pathological examination of the surgical samples indicated a variety of destructive changes in addition to ongoing inflammation. The gross specimen of S1 and S2 segments of the left lung showed fibrocavernous tuberculosis shown in Fig. 4 B, which corresponds to the left lung lesion seen on the first preoperative CT in Figs. 3 B and 5 A in the second preoperative CT; the gross specimen of the S6 segment showed progressive tuberculoma seen in Fig. 4 C, which corresponds to the left lung lesion seen on the first preoperative CT in Figs. 3 D and 5 C in the second preoperative CT.
There were no postoperative complications, and tuberculosis (TB) treatment was reinitiated. The patient successfully completed treatment with normalization of clinical and laboratory parameters and a clinical outcome of cure in September 2021, ~ 14 months after beginning treatment. The patient had reported near complete resolution of her symptoms, having a much better ability to perform her daily activities. The patient appreciated the effects surgery had on her recovery and was happy to have gone through that treatment route. A post treatment CT scan demonstrated postoperative changes in the upper segments of both lungs (Fig. 5 D). Results from post treatment lung function testing were all within normal range.
Discussion and conclusions
We present this case to highlight the heterogeneous nature of pulmonary tuberculosis and need for an individualized treatment approach, especially for patients with cavitary disease. Over the last decade, novel diagnostics, drugs, and treatment regimens have revolutionized TB management including a recent landmark clinical trial demonstrating an effective 4-month regimen for drug-susceptible TB [ 1 ]. The move towards shorter regimens is critical to improve treatment completion rates and help meet TB elimination goals. However, during a transition to shorter treatment durations it is imperative that clinicians remain aware of complex and severe pulmonary TB cases that may require longer durations of treatment and adjunctive therapies such as surgery. Supporting evidence comes from a recent landmark study finding persistent inflammation on imaging associated with finding Mtb mRNA in sputum after successful treatment and a meta-analysis demonstrating a hard-to-treat TB phenotype not cured with the standard 6 months of treatment [ 2 , 5 ]. However, regarding recommendations for prolonging treatment beyond 6 months for drug-susceptible pulmonary tuberculosis, ATS/CDC/IDSA recommends (expert opinion) extended treatment for persons with cavitary disease and a positive 2 month culture (our patient would not have met this criteria); World Health Organization (WHO) does not recommend extended treatment for any persons with drug-susceptible TB [ 6 , 7 ]. Accumulating evidence demonstrates surgical resection may be an effective adjunctive treatment in cases with cavitary disease [ 8 , 9 , 10 , 11 , 12 ]. Ultimately, a precision medicine approach towards TB will be able to identify patients who would benefit from short course therapy and those who would benefit from longer therapy and adjunctive treatment including surgery [ 13 ].
Mtb has a unique ability and propensity to induce cavities in humans with various studies showing cavitary lesions in ~ 30 to 85% of patients with pulmonary tuberculosis [ 14 ]. Lung cavities are more common in certain groups including patients with diabetes mellitus and undernutrition such as our patient who had a baseline BMI of 16.3 kg/m 2 [ 15 , 16 ]. Their presence indicates more advanced and severe pulmonary disease as evidenced by their association with worse clinical outcomes. Cavitary disease has been associated with higher rates of treatment failure, disease relapse, acquired drug resistance, and long term-term pulmonary morbidity [ 2 , 17 , 18 , 19 ]. The impact of cavitary disease may be more pronounced in drug-resistant disease as shown in an observational study from our group which found a five times higher rate of acquired drug resistance and eight times higher rate of treatment failure among patients multidrug- or extensively drug-resistant cavitary disease compared to those without [ 20 ].
Mtb cavities are characterized by a fibrotic surface with variable vascularization, a lymphocytic cuff at the periphery followed by a cellular layer consisting of primarily macrophages and a necrotic center with foamy apoptotic macrophages and high concentrations of bacteria. Historically, each portion of the TB cavity has been conceptualized as concentric layers of a spherical structure due to its appearance on histologic cross-sections. However, recent studies using more detailed imaging techniques have shown most TB cavities exhibit complex structures with diverse, branching morphologies [ 21 ]. A dysregulated host immune response to Mtb is thought to contribute to the development of lung cavities, which may explain why cavitary lesions are seen less frequently among immunosuppressed patients including people living with Human Immunodeficiency Virus (HIV) [ 14 ]. The center of the TB cavity (caseum) is characterized by accumulation of pro-inflammatory lipid signaling molecules (eicosanoids) and reactive oxygen species, which result in ongoing tissue destruction, but do little to control Mtb replication [ 22 ]. Conversely, the cellular rim and lymphocytic cuff are characterized by a lower abundance of pro-inflammatory lipids and increases in immunosuppressive signals including elevated expression of TGF-beta and indoleamine-2,3-dioxygenase-1 [ 22 ]. The anti-inflammatory milieu within these TB cavity microenvironments impairs effector T cell responses, further limiting control of bacterial replication [ 23 , 24 , 25 ].
The combination of impaired cell-mediated immune responses with accumulation of inflammatory mediators at the rim of the caseum leads to ongoing tissue destruction with the potential for long-term pulmonary sequelae. Many with cavitary tuberculosis suffer chronic obstructive pulmonary disease after successful treatment and the risk may be greater in those with multidrug-resistant disease [ 3 , 4 ]. This has led to research into adjunctive treatment with immune modulator therapies with a goal of mitigating the over-exuberant inflammatory response at the interior edge of the cavity to limit tissue damage. In a recent randomized clinical trial, patients with radiographically severe pulmonary tuberculosis treated with adjunctive everolimus or CC-11050 (phosphodiesterase inhibitor with anti-inflammatory properties) achieved better long-term pulmonary outcomes versus those who received placebo [ 26 ]. Such results suggest the inflammatory response can be modified with appropriate host-directed therapies to improve pulmonary outcomes, particularly in those with cavitary tuberculosis.
Tuberculosis cavities not only hinder an effective immune response, but also prevent anti-tuberculosis drugs from achieving sterilizing concentrations throughout the lesion and especially in necrotic regions. The necrotic center of cavitary lesions is associated with extremely high rates of bacilli (up to 10 9 per milliliter), many of which enter a dormant state with reduced metabolic activity. Bacilli in this dormant state may be less responsive to the host immune response and exhibit phenotypic resistance to some anti-tuberculosis drugs thereby preventing sterilization and increasing chances of relapse [ 14 , 27 , 28 ]. The fact that the specimens from our patient’s second surgery were Xpert and AFB positive, but culture negative may indicate the presence of either dead bacilli or metabolically altered(dormant) bacilli that may be alive, but not culturable by standard techniques. Further, genomic sequencing studies have also found distinct strains of Mtb within different areas of the cavity that have varying drug-susceptibilities demonstrating cavities as a potential incubator for drug resistance [ 27 , 29 ].
Emerging literature has started to elucidate the varying abilities of drugs to penetrate into cavitary lesions and the importance of adequate target site concentrations. One notable study found that decreasing tissue concentrations within resected cavitary TB lesions were associated with increasing drug phenotypic MIC values [ 30 ]. Innovative studies using MALDI mass spectrometry imaging have further demonstrated varied spatiotemporal penetration of anti-TB drugs in human TB cavities [ 31 ]. This study found rifampin accumulated within caseum, moxifloxacin preferentially at the cellular rim, and pyrazinamide throughout the lesion, demonstrating the need to consider drug penetration when designing drug regimens in patients with cavitary TB. Computational modeling studies have further demonstrated the importance of complete lesion drug coverage to ensure relapse-free cure [ 32 ]. Furthermore, clinical trials are now incorporating these principles into study design by (1) using radiological characteristics to determine treatment length and (2) incorporating tissue penetration into drug selection and regimen design [ 33 , 34 ]. Beyond tissue penetration, varying drug levels and rapid INH acetylation status can also lead to suboptimal pharmacokinetics and poor clinical outcomes [ 35 , 36 ]. As highlighted in a recent expert document, clinical standards to optimize and individualize dosing need to be developed to improve outcomes [ 37 ].
Available literature points to a benefit of adjunctive surgical resection particularly among patients with drug resistant tuberculosis. A meta-analysis of 24 comparative studies found surgical intervention was associated with favorable treatment outcomes among patients with drug-resistant TB (odds ratio 2.24, 95% CI 1.68–2.97) [ 38 ]. Additionally, an individual patient data meta-analysis found that partial lung resection (adjusted OR 3.9, 95% CI 1.5–5.9) but not pneumectomy was associated with treatment success [ 39 ]. In two observational studies, we have also found that adjunctive surgical resection was associated with high and improved outcomes compared to patients with cavitary disease not undergoing surgery and was associated with less reentry into TB care. It should be noted that all studies of surgical resection for pulmonary TB were observational studies, which may be subject to selection bias, and no clinical trials (very difficult to implement in practice) were conducted to provide more conclusive evidence. Based on available evidence, the WHO has provided guidance to consider surgery among certain hard to treat cases of both drug-susceptible and resistant cavitary disease [ 40 ]. Criteria for surgical intervention included (1) failure of medical therapy (persistent sputum culture positive for M. tuberculosis ), (2) a high likelihood of treatment failure or disease relapse, (3) complications from the disease, (4) localized cavitary lesion, and (5) sufficient pulmonary function to tolerate surgery. For our patient, the severity of disease, lack of improvement of radiological imaging despite appropriate treatment, and high risk of relapse were the main indicators for surgery. Contraindications for surgery included a forced expiratory volume (FEV1) < 1000 mL, severe malnutrition, or patients at high risk for perioperative cardiovascular complications. With strict adherence to indications and contraindications for surgery, an acceptable level of postoperative complications are noted (5–17%) [ 4 , 38 ]. Our results also demonstrate the safety of adjunctive surgery, as our post-operative complication rate (8%) was low with the majority being minor complications [ 41 ].
As our case highlights, patients with persistent cavitary disease at the end of treatment require close clinical follow up and a tailored, individualized plan to determine the best approach for disease elimination and cure. In certain cases, including those with persistent cavitary disease and end of treatment, and where available, surgical resection is an effective adjunctive treatment option that can reduce disease burden and aid anti-tuberculosis agents in providing a sterilizing cure. As we enter an era of welcomed new shorter treatment options for tuberculosis it is imperative for clinicians to be able to identify and recognize complicated TB cases that require prolonged treatment and potentially adjunctive surgery.
Availability of data and materials
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Abbreviations
Acid fast bacilli
American Thoracic Society
Body mass index
Center for Disease Control
Computed tomography
Directly observed therapy
Drug sensitive tuberculosis
Erythrocyte sedimentation rate
Human Immunodeficiency Virus
Infectious Diseases Society of America
Mycobacterium tuberculosis
- Tuberculosis
World Health Organization
Dorman S, Nahid P, Kurbatova E, Phillips P, Bryant K, Dooley K, et al. Four-month rifapentine regimens with or without moxifloxacin for tuberculosis. N Engl J Med. 2021;384(18):1705–18.
Article PubMed PubMed Central CAS Google Scholar
Imperial M, Nahid P, Phillips P, Davies G, Fielding K, Hanna D, et al. A patient-level pooled analysis of treatment-shortening regimens for drug-susceptible pulmonary tuberculosis. Nat Med. 2018;24(11):1708–15.
Vashakidze S, Kempker J, Jakobia N, Gogishvili S, Nikolaishvili K, Goginashvili L, et al. Pulmonary function and respiratory health after successful treatment of drug-resistant tuberculosis. Int J Infect Dis. 2019;82:66–72.
Article PubMed PubMed Central Google Scholar
Harris RC, Khan MS, Martin LJ, et al. The effect of surgery on the outcome of treatment for multidrug-resistant tuberculosis: a systematic review and meta-analysis. BMC Infect Dis. 2016;16:262.
Malherbe S, Shenai S, Ronacher K, Loxton A, Dolganov G, Kriel M, et al. Persisting positron emission tomography lesion activity and Mycobacterium tuberculosis mRNA after tuberculosis cure. Nat Med. 2016;22(10):1094–100.
Nahid P, Dorman S, Alipanah N, Barry P, Brozek J, Cattamanchi A, et al. Official American Thoracic Society/Centers for Disease Control and Prevention/Infectious Diseases Society of America clinical practice guidelines: treatment of drug-susceptible tuberculosis. Clin Infect Dis. 2016;63(7):e147–95.
World Health Organization. The WHO consolidated guidelines on tuberculosis, module 4: treatment—drug-susceptible tuberculosis treatment. Geneva: WHO; 2022.
Google Scholar
Kang M, Kim H, Choi Y, Kim K, Shim Y, Koh W, et al. Surgical treatment for multidrug-resistant and extensive drug-resistant tuberculosis. Ann Thorac Surg. 2010;89(5):1597–602.
Article PubMed Google Scholar
Pomerantz B, Cleveland J, Olson H, Pomerantz M. Pulmonary resection for multi-drug resistant tuberculosis. J Thorac Cardiovasc Surg. 2001;121(3):448–53.
Article PubMed CAS Google Scholar
Somocurcio J, Sotomayor A, Shin S, Portilla S, Valcarcel M, Guerra D, et al. Surgery for patients with drug-resistant tuberculosis: report of 121 cases receiving community-based treatment in Lima, Peru. Thorax. 2007;62(5):416–21.
Dravniece G, Cain K, Holtz T, Riekstina V, Leimane V, Zaleskis R. Adjunctive resectional lung surgery for extensively drug-resistant tuberculosis. Eur Respir J. 2009;34(1):180–3.
Wang H, Lin H, Jiang G. Pulmonary resection in the treatment of multidrug-resistant tuberculosis: a retrospective study of 56 cases. Ann Thorac Surg. 2008;86(5):1640–5.
Lange C, Aarnoutse R, Chesov D, van Crevel R, Gillespie S, Grobbel H, et al. Perspective for precision medicine for tuberculosis. Front Immunol. 2020. https://doi.org/10.3389/fimmu.2020.566608 .
Urbanowski M, Ordonez A, Ruiz-Bedoya C, Jain S, Bishai W. Cavitary tuberculosis: the gateway of disease transmission. Lancet Infect Dis. 2020;20(6):e117–28.
Zafar M, Chen L, Xiaofeng Y, Gao F. Impact of diabetes mellitus on radiological presentation of pulmonary tuberculosis in otherwise non-immunocompromised patients: a systematic review. Curr Med Imaging Rev. 2019;15(6):543–54.
Sinha P, Bhargava A, Carwile M, Cintron C, Cegielski J, Lönnroth K, et al. Undernutrition can no longer be an afterthought for global efforts to eliminate TB. Int J Tuberc Lung Dis. 2022;26(6):477–80.
Cegielski J, Dalton T, Yagui M, Wattanaamornkiet W, Volchenkov G, Via L, et al. Extensive drug resistance acquired during treatment of multidrug-resistant tuberculosis. Clin Infect Dis. 2014;59(8):1049–63.
Gao J, Ma Y, Du J, Zhu G, Tan S, Fu Y, et al. Later emergence of acquired drug resistance and its effect on treatment outcome in patients treated with Standard Short-Course Chemotherapy for tuberculosis. BMC Pulm Med. 2016. https://doi.org/10.1186/s12890-016-0187-3 .
Shin S, Keshavjee S, Gelmanova I, Atwood S, Franke M, Mishustin S, et al. Development of extensively drug-resistant tuberculosis during multidrug-resistant tuberculosis treatment. Am J Respir Crit Care Med. 2010;182(3):426–32.
Kempker R, Kipiani M, Mirtskhulava V, Tukvadze N, Magee M, Blumberg H. Acquired drug resistance in Mycobacterium tuberculosis and poor outcomes among patients with multidrug-resistant tuberculosis. Emerg Infect Dis. 2015;21(6):992–1001.
Wells G, Glasgow J, Nargan K, Lumamba K, Madansein R, Maharaj K, et al. Micro-computed tomography analysis of the human tuberculous lung reveals remarkable heterogeneity in three-dimensional granuloma morphology. Am J Respir Crit Care Med. 2021;204(5):583–95.
Marakalala M, Raju R, Sharma K, Zhang Y, Eugenin E, Prideaux B, et al. Inflammatory signaling in human tuberculosis granulomas is spatially organized. Nat Med. 2016;22(5):531–8.
McCaffrey E, Donato M, Keren L, Chen Z, Delmastro A, Fitzpatrick M, et al. The immunoregulatory landscape of human tuberculosis granulomas. Nat Immunol. 2022;23(2):318–29.
Gern B, Adams K, Plumlee C, Stoltzfus C, Shehata L, Moguche A, et al. TGFβ restricts expansion, survival, and function of T cells within the tuberculous granuloma. Cell Host Microbe. 2021;29(4):594-606.e6.
Gautam U, Foreman T, Bucsan A, Veatch A, Alvarez X, Adekambi T, et al. In vivo inhibition of tryptophan catabolism reorganizes the tuberculoma and augments immune-mediated control of Mycobacterium tuberculosis. Proc Natl Acad Sci USA. 2017. https://doi.org/10.1073/pnas.1711373114 .
Wallis R, Ginindza S, Beattie T, Arjun N, Likoti M, Edward V, et al. Adjunctive host-directed therapies for pulmonary tuberculosis: a prospective, open-label, phase 2, randomised controlled trial. Lancet Respir Med. 2021;9(8):897–908.
Kaplan G, Post F, Moreira A, Wainwright H, Kreiswirth B, Tanverdi M, et al. Mycobacterium tuberculosis growth at the cavity surface: a microenvironment with failed immunity. Infect Immun. 2003;71(12):7099–108.
Fattorini L, Piccaro G, Mustazzolu A, Giannoni F. Targeting dormant bacilli to fight tuberculosis. Mediterr J Hematol Infect Dis. 2013;5(1):e2013072.
Moreno-Molina M, Shubladze N, Khurtsilava I, Avaliani Z, Bablishvili N, Torres-Puente M, et al. Genomic analyses of Mycobacterium tuberculosis from human lung resections reveal a high frequency of polyclonal infections. Nat Commun. 2021;12(1):2716.
Dheda K, Lenders L, Magombedze G, Srivastava S, Raj P, Arning E, et al. Drug-penetration gradients associated with acquired drug resistance in patients with tuberculosis. Am J Respir Crit Care Med. 2018;198(9):1208–19.
Prideaux B, Via L, Zimmerman M, Eum S, Sarathy J, O’Brien P, et al. The association between sterilizing activity and drug distribution into tuberculosis lesions. Nat Med. 2015;21(10):1223–7.
Strydom N, Gupta S, Fox W, Via L, Bang H, Lee M, et al. Tuberculosis drugs’ distribution and emergence of resistance in patient’s lung lesions: a mechanistic model and tool for regimen and dose optimization. PLoS Med. 2019;16(4): e1002773.
Chen R, Via L, Dodd L, Walzl G, Malherbe S, Loxton A, et al. Using biomarkers to predict TB treatment duration (predict TB): a prospective, randomized, noninferiority, treatment shortening clinical trial. Gates Open Res. 2017;1:9.
Bartelink I, Zhang N, Keizer R, Strydom N, Converse P, Dooley K, et al. New paradigm for translational modeling to predict long-term tuberculosis treatment response. Clin Transl Sci. 2017;10(5):366–79.
Pasipanodya J, Srivastava S, Gumbo T. Meta-analysis of clinical studies supports the pharmacokinetic variability hypothesis for acquired drug resistance and failure of antituberculosis therapy. Clin Infect Dis. 2012;55(2):169–77.
Colangeli R, Jedrey H, Kim S, Connell R, Ma S, Chippada Venkata U, et al. Bacterial factors that predict relapse after tuberculosis therapy. N Engl J Med. 2018;379(9):823–33.
Alffenaar J, Stocker S, Forsman L, Garcia-Prats A, Heysell S, Aarnoutse R, et al. Clinical standards for the dosing and management of TB drugs. Int J Tuberc Lung Dis. 2022;26(6):483–99.
Marrone M, Venkataramanan V, Goodman M, Hill A, Jereb J, Mase S. Surgical interventions for drug-resistant tuberculosis: a systematic review and meta-analysis [review article]. Int J Tuberc Lung Dis. 2013;17(1):6–16.
Fox G, Mitnick C, Benedetti A, Chan E, Becerra M, Chiang C, et al. Surgery as an adjunctive treatment for multidrug-resistant tuberculosis: an individual patient data metaanalysis. Clin Infect Dis. 2016;62(7):887–95.
Word Health Organization Europe. The role of surgery in the treatment of pulmonary TB and multidrug and extensively drug-resistant TB. Copenhagen: WHO Regional Office for Europe; 2014.
Vashakidze SA, Gogishvili SG, Nikolaishvili KG, et al. Adjunctive surgery versus medical treatment among patients with cavitary multidrug-resistant tuberculosis. Eur J Cardiothorac Surg. 2021;60(6):1279–85. https://doi.org/10.1093/ejcts/ezab337 .
Download references
Acknowledgements
The authors thank the physicians, nurses, and staff at the NCTLD in Tbilisi, Georgia, who provided care for the patient described in this report. Additionally, the authors are thankful for the patient with pulmonary tuberculosis who was willing to have their course of illness presented and help contribute meaningful data that may help future patients with the same illness.
This study did not receive any specific funding.
Author information
Authors and affiliations.
Thoracic Surgery Department, National Center for Tuberculosis and Lung Diseases, 50 Maruashvili, 0101, Tbilisi, Georgia
Sergo A. Vashakidze, Merab Japaridze, Giorgi Gogishvili & Manana Rekhviashvili
The University of Georgia, Tbilisi, Georgia
Sergo A. Vashakidze
Tbilisi State Medical University, Tbilisi, Georgia
Abivarma Chandrakumaran
Division of Infectious Diseases, Department of Medicine, Emory University School of Medicine, Atlanta, GA, USA
Jeffrey M. Collins & Russell R. Kempker
You can also search for this author in PubMed Google Scholar
Contributions
SAV: Conceptualization; Data collection and interpretation; Scientific Writing including initial draft preparation and manuscript revision and editing. AC: Data interpretation; Table and Figure preparation; Literature review; Scientific Writing including initial draft preparation and manuscript revision and editing. MJ: Data collection; Scientific Writing including manuscript review and editing. GG: Data collection; Scientific Writing including manuscript review and editing. JMC: Data interpretation; Scientific Writing including manuscript review and editing. MR: Data interpretation; Scientific Writing including manuscript review and editing. RRK: Conceptualization; Literature review; Scientific Writing including manuscript review and editing. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Sergo A. Vashakidze .
Ethics declarations
Ethics approval and consent to participate.
The authors confirm that written informed consent has been obtained from the patient involved in the case report. Ethics approval is not needed for case reports according to our institutional (National Center for Tuberculosis and Lung Disease) review board.
Consent for publication
The authors confirm that written consent has been obtained from the patient for publication of images.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Vashakidze, S.A., Chandrakumaran, A., Japaridze, M. et al. A case report of persistent drug-sensitive pulmonary tuberculosis after treatment completion. BMC Infect Dis 22 , 864 (2022). https://doi.org/10.1186/s12879-022-07836-y
Download citation
Received : 08 August 2022
Accepted : 02 November 2022
Published : 19 November 2022
DOI : https://doi.org/10.1186/s12879-022-07836-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Thoracic surgery
BMC Infectious Diseases
ISSN: 1471-2334
- General enquiries: [email protected]
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
A case study of a patient with multidrug-resistant tuberculosis
Affiliation.
- 1 Community nurse working in South West England.
- PMID: 30048191
- DOI: 10.12968/bjon.2018.27.14.806
In this case study, a nurse presents her reflections on the challenges of supporting a patient through his treatment journey for multidrug-resistant tuberculosis. The patient has significant comorbidities and social issues, such as diabetes and homelessness. There was also a language barrier. All these aspects made the management of his treatment challenging. The medication side effects and his lifestyle were also a barrier to full engagement. The same multidisciplinary team was involved with the patient and, despite the obstacles, he seemed willing to engage with treatment and the team.
Keywords: Comorbidities; Language barrier; Multidisciplinary team; Multidrug-resistant tuberculosis; Pulmonary TB; Under-served population.
PubMed Disclaimer
Similar articles
- I know how TB patients feel. Burden M. Burden M. Nurs Stand. 2016 Nov 16;31(12):29. doi: 10.7748/ns.31.12.29.s27. Nurs Stand. 2016. PMID: 27897750
- The role of the nurse in the community-based treatment of multidrug-resistant tuberculosis (MDR-TB). Palacios E, Guerra D, Llaro K, Chalco K, Sapag R, Furin J. Palacios E, et al. Int J Tuberc Lung Dis. 2003 Apr;7(4):343-6. Int J Tuberc Lung Dis. 2003. PMID: 12729339
- Off-Label Use of Bedaquiline in Children and Adolescents with Multidrug-Resistant Tuberculosis. Achar J, Hewison C, Cavalheiro AP, Skrahina A, Cajazeiro J, Nargiza P, Herboczek K, Rajabov AS, Hughes J, Ferlazzo G, Seddon JA, du Cros P. Achar J, et al. Emerg Infect Dis. 2017 Oct;23(10):1711-3. doi: 10.3201/eid2310.170303. Epub 2017 Oct 17. Emerg Infect Dis. 2017. PMID: 28758889 Free PMC article.
- Peripheral neuropathy in a diabetic child treated with linezolid for multidrug-resistant tuberculosis: a case report and review of the literature. Swaminathan A, du Cros P, Seddon JA, Mirgayosieva S, Asladdin R, Dusmatova Z. Swaminathan A, et al. BMC Infect Dis. 2017 Jun 12;17(1):417. doi: 10.1186/s12879-017-2499-1. BMC Infect Dis. 2017. PMID: 28606115 Free PMC article. Review.
- Treatment correlates of successful outcomes in pulmonary multidrug-resistant tuberculosis: an individual patient data meta-analysis. Collaborative Group for the Meta-Analysis of Individual Patient Data in MDR-TB treatment–2017; Ahmad N, Ahuja SD, Akkerman OW, Alffenaar JC, Anderson LF, Baghaei P, Bang D, Barry PM, Bastos ML, Behera D, Benedetti A, Bisson GP, Boeree MJ, Bonnet M, Brode SK, Brust JCM, Cai Y, Caumes E, Cegielski JP, Centis R, Chan PC, Chan ED, Chang KC, Charles M, Cirule A, Dalcolmo MP, D'Ambrosio L, de Vries G, Dheda K, Esmail A, Flood J, Fox GJ, Fréchet-Jachym M, Fregona G, Gayoso R, Gegia M, Gler MT, Gu S, Guglielmetti L, Holtz TH, Hughes J, Isaakidis P, Jarlsberg L, Kempker RR, Keshavjee S, Khan FA, Kipiani M, Koenig SP, Koh WJ, Kritski A, Kuksa L, Kvasnovsky CL, Kwak N, Lan Z, Lange C, Laniado-Laborín R, Lee M, Leimane V, Leung CC, Leung EC, Li PZ, Lowenthal P, Maciel EL, Marks SM, Mase S, Mbuagbaw L, Migliori GB, Milanov V, Miller AC, Mitnick CD, Modongo C, Mohr E, Monedero I, Nahid P, Ndjeka N, O'Donnell MR, Padayatchi N, Palmero D, Pape JW, Podewils LJ, Reynolds I, Riekstina V, Robert J, Rodriguez M, Seaworth B, Seung KJ, Schnippel K, Shim TS, Singla R, Smith SE, Sotgiu G, Sukhbaatar G, Tabarsi P, Tiberi S, Trajman A, Trieu L, Udwadia ZF, van der Werf TS, Veziris N, Viiklepp P, Vilbrun SC,… See abstract for full author list ➔ Collaborative Group for the Meta-Analysis of Individual Patient Data in MDR-TB treatment–2017, et al. Lancet. 2018 Sep 8;392(10150):821-834. doi: 10.1016/S0140-6736(18)31644-1. Lancet. 2018. PMID: 30215381 Free PMC article. Review.
- Search in MeSH
Related information
Linkout - more resources, full text sources, other literature sources.
- scite Smart Citations
- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
- Fact sheets
- Facts in pictures
- Publications
- Questions and answers
- Tools and toolkits
- Endometriosis
- Excessive heat
- Mental disorders
- Polycystic ovary syndrome
- All countries
- Eastern Mediterranean
- South-East Asia
- Western Pacific
- Data by country
- Country presence
- Country strengthening
- Country cooperation strategies
- News releases
- Feature stories
- Press conferences
- Commentaries
- Photo library
- Afghanistan
- Cholera
- Coronavirus disease (COVID-19)
- Greater Horn of Africa
- Israel and occupied Palestinian territory
- Disease Outbreak News
- Situation reports
- Weekly Epidemiological Record
- Surveillance
- Health emergency appeal
- International Health Regulations
- Independent Oversight and Advisory Committee
- Classifications
- Data collections
- Global Health Observatory
- Global Health Estimates
- Mortality Database
- Sustainable Development Goals
- Health Inequality Monitor
- Global Progress
- World Health Statistics
- Partnerships
- Committees and advisory groups
- Collaborating centres
- Technical teams
- Organizational structure
- Initiatives
- General Programme of Work
- WHO Academy
- Investment in WHO
- WHO Foundation
- External audit
- Financial statements
- Internal audit and investigations
- Programme Budget
- Results reports
- Governing bodies
- World Health Assembly
- Executive Board
- Member States Portal

Section navigation
- Featured topics
- Country case studies
- International donor funding
- New treatment for TB
- TB in prisons
- UN declaration on TB
Notable findings from two recent TB epidemiological reviews in the WHO African Region
Epidemiological reviews of tuberculosis (TB) comprise an evaluation of the capacity of the surveillance system to directly measure the number of TB cases and deaths, using the World Health Organization (WHO) surveillance checklist of standards and benchmarks ( 1 ), coupled with an epidemiological analysis to assess the level of, and trends in, TB burden in the country. Such reviews have four main components: analysis of TB (and other relevant) data, interviews with national TB programmes (NTPs) and relevant stakeholders, desk reviews of documents, and health facility visits to understand people and data flows. Findings and recommendations from these reviews are used to inform programme reviews and strategic planning activities; they are also used in developing concept notes for domestic and international funding requests. Standardized terms of reference (ToR) for TB epidemiological reviews have been available since 2013, with the latest being the 2018 ToR ( 2 ). Key findings and recommendations made to NTPs from epidemiological reviews carried out in the high TB burden countries have been synthesized ( 3 ).
This document gives examples of notable findings from two countries: Nigeria and the United Republic of Tanzania.
Nigeria, January 2023
Nigeria conducted an epidemiological review on 9–20 January 2023, its fourth since 2014. The time trend in TB case notifications since 2014 has been fairly stable, at around 50 cases per 100 000 population Fig. 1 . Findings from the 2020 epidemiological review suggested that the most important factor for this low level of case notification and the corresponding low levels of treatment coverage (24% in 2018) was the underdiagnosis of people with TB, which in turn was due to issues with the network of primary health care (PHC) facilities. The underdiagnosis was compounded by the underreporting of people with TB, primarily from the private sector and particularly in urban settings.
Four major routine programmatic activities were introduced around 2020 to address these findings, increase screening coverage and strengthen diagnosis and reporting of people with TB:
- active screening in the outpatient departments (OPD) of PHCs, where all those attending OPD are screened for TB symptoms upon registration and are re-routed through TB services for sputum collection and testing if screening is positive;
- expansion of TB service provision through further engagement of the PHC network, with the provision of additional GeneXpert machines;
- engagement with private service providers to provide diagnosis, treatment and reporting of people with TB; and
- community outreach activities, including home-based screening and referral of presumptive TB cases to a local health facility.
The 2023 epidemiological review found excellent progress in case finding and reporting, with the absolute number of total case notifications increasing from 103 018 in 2018 to 285 561 in 2022, corresponding to an increase in estimated treatment coverage from 24% in 2018 to 59% in 2022 Fig. 1 . Among the 282 184 new and relapse TB cases notified in 2022, 99% were pulmonary, of which 81% were bacteriologically confirmed. Despite the sharp increase in case notifications, the NTP maintained the high quality of services offered in 2022: treatment success was 91%, and 97% of new and relapse TB cases had a documented HIV status at diagnosis.
The NTP remained resilient to the shock of the coronavirus disease (COVID-19) pandemic and only experienced a small decrease in case notifications during a lockdown in the second quarter of 2020. In fact, the implementation of parallel TB and COVID-19 screening during the height of the pandemic contributed to increased TB case finding.
Although the gains made in Nigeria are obvious, an important gap remains in finding all people living with TB. This is due to various factors, some of which are clearly the responsibility of the NTP (e.g. incomplete TB service coverage of PHCs) whereas others are not so strongly linked to the NTP (e.g. barriers to universal health coverage). Therefore, increased investment will be needed to maintain momentum and continue expanding these successful programmatic strategies. As case detection is scaled up further, there is a risk that the system in place for caring for the patients will become saturated and the quality of care may drop. Therefore, investment will also need to be strengthened along the rest of the pathway of prevention and care, to ensure that capacity and resources are sufficient to maintain high-quality and timely laboratory testing and transmission of results, high treatment success rates, high coverage of HIV testing among diagnosed TB cases, and recording and reporting of high-quality data.
Fig. 1 Notifications of TB cases – overall, new and relapse all forms of TB, pulmonary bacteriologically confirmed and clinically diagnosed, 2014–2022, Nigeria
United republic of tanzania, january 2023.
The United Republic of Tanzania conducted an epidemiological review on 15–25 January 2023, its third since 2014. The number of TB case notifications was relatively constant over the period 2001–2015 (between 60 000 and 65 000 cases) and then increased slowly until 2018, where it reached a peak of more than 70 000 TB cases notified Fig. 2 . In 2019, a 15% increase was seen compared with 2018, and a further increase of 15% was seen between 2021 and 2022, where almost 100 000 TB cases were notified.
The increase in TB notification since 2018 was mainly due to an increase in clinically diagnosed TB, although the number of laboratory-confirmed TB cases remained constant over time. Another shift was observed in 2022, with the number of clinically diagnosed TB increasing much faster than the number of bacteriologically confirmed TB. Upon further investigation and analysis during the epidemiological review, this increase could be partly explained by the national screening and diagnosis algorithm, which recommends that presumptive TB cases with a negative Xpert result undergo a clinical assessment and chest radiography. If the outcome of this assessment is “suggestive of TB”, patients start TB treatment, despite the negative bacteriological evidence. At the national level, it has been shown that about a third of people with a negative Xpert result still start TB treatment based on the clinical assessment and chest radiography. However, it is not clear whether “suggestive for TB” is a sufficiently specific outcome to treat people for TB among a population with high transmission and hence high prevalence of lung abnormalities (e.g. “healed” TB). The review recommended further and more regular analyses of case-based data, and a small-scale study to review a sample of medical records and re-read chest X-rays, to rule out overdiagnosis caused by the current algorithm.
The TB programme made significant progress in 2018 with the establishment, and subsequent national roll-out, of a digital case-based surveillance system for TB (ETL/DHIS2). The system was last updated in September 2021 and is supported by a strong national team. However, other parts of the Ministry of Health and other parts of government are responsible for the development of a national digital strategy, as well as the development and roll-out of new digital health data systems. The NTP needs to strengthen collaboration and coordination with these government agencies, to ensure its needs are met in terms of surveillance, monitoring and evaluation, and other TB-related administrative processes. This could include the establishment of a national technical group to discuss key issues such as the establishment of a unique identification (ID) for health, data linkages with laboratories, and routine analysis and use of data at all levels including that of the health facility.
Fig. 2 Notifications of TB cases – overall, new and relapse all forms of TB, pulmonary bacteriologically confirmed and clinically diagnosed, 2001–2022, United Republic of Tanzania
WHO consolidated guidelines on tuberculosis data generation and use. Module 1: Tuberculosis surveillance. Web Annex B. Standards and benchmarks for tuberculosis surveillance and vital registration systems: checklist (2nd edition). Geneva: World Health Organization; (In press).
Assessing the performance of national TB surveillance using the WHO TB surveillance checklist of standards and benchmarks, 2013–2020: synthesis of findings from high TB burden countries. Geneva: World Health Organization; (In press).
Guidance on conducting reviews of tuberculosis programmes. Geneva: World Health Organization; (In press).
- Open access
- Published: 21 February 2023
Tuberculosis in older adults: case studies from four countries with rapidly ageing populations in the western pacific region
- Alvin Kuo Jing Teo 1 , 2 , 11 ,
- Kalpeshsinh Rahevar 3 ,
- Fukushi Morishita 3 ,
- Alicia Ang 15 ,
- Takashi Yoshiyama 5 ,
- Akihiro Ohkado 5 ,
- Lisa Kawatsu 5 ,
- Norio Yamada 5 ,
- Kazuhiro Uchimura 5 ,
- Youngeun Choi 6 ,
- Zi Chen 7 ,
- Siyan Yi 1 , 8 , 9 ,
- Manami Yanagawa 3 ,
- Kyung Hyun Oh 3 ,
- Kerri Viney 10 na1 ,
- Ben Marais 2 , 11 na1 ,
- Heejin Kim 6 na1 ,
- Seiya Kato 5 na1 ,
- Yuhong Liu 12 na1 ,
- Catherine W.M. Ong 4 , 13 , 14 na1 &
- Tauhid Islam 3 na1
BMC Public Health volume 23 , Article number: 370 ( 2023 ) Cite this article
4826 Accesses
10 Citations
1 Altmetric
Metrics details
A Correction to this article was published on 07 June 2023
This article has been updated
The Western Pacific Region has one of the fastest-growing populations of older adults (≥ 65 years) globally, among whom tuberculosis (TB) poses a particular concern. This study reports country case studies from China, Japan, the Republic of Korea, and Singapore reflecting on their experiences in managing TB among older adults.
Across all four countries, TB case notification and incidence rates were highest among older adults, but clinical and public health guidance focused on this population was limited. Individual country reports illustrated a range of practices and challenges. Passive case finding remains the norm, with limited active case finding (ACF) programs implemented in China, Japan, and the Republic of Korea. Different approaches have been trialled to assist older adults in securing an early diagnosis, as well as adhering to their TB treatment. All countries emphasised the need for person-centred approaches that include the creative application of new technology and tailored incentive programs, as well as reconceptualisation of how we provide treatment support. The use of traditional medicines was found to be culturally entrenched among older adults, with a need for careful consideration of their complementary use. TB infection testing and the provision of TB preventive treatment (TPT) were underutilised with highly variable practice.
Older adults require specific consideration in TB response policies, given the burgeoning aging population and their high TB risk. Policymakers, TB programs and funders must invest in and develop locally contextualised practice guidelines to inform evidence-based TB prevention and care practices for older adults.
Peer Review reports
Globally the number of older adults (aged ≥ 65 years) is expected to triple by 2100 [ 1 ]. Life expectancy at birth has gradually increased since the 1950s, [ 1 ] with the number of older adults projected to exceed children aged < 5, across all economies by 2020 [ 2 ]. The Western Pacific Region has one of the fastest-growing populations of older adults globally. In 2019, life expectancy at birth of the population was 4 years above the global estimate of 73.3 years [ 3 ]. Japan, for instance, is the most aged country in the world (average life expectancy 85 years) [ 4 ]. Several other countries in the region, particularly the People’s Republic of China (hereinafter referred to as China), the Republic of Korea, and the Republic of Singapore (hereinafter referred to as Singapore), were estimated to have an increase in the proportion of older adults globally between 2019 and 2050.
Japan has the highest proportion of older adults in the world, at 28% of the total population in 2020, [ 5 ] and the proportion is expected to rise to 38% by 2065 [ 6 ]. China is also undergoing a profound demographic transition. In 2021, the number of older adults reached 191 million, amounting to 13.5% of the total population. The number rose by 4.6% compared to the previous year, [ 7 ] and the increasing trend is likely to persist with the number of older adults expected to exceed 480 million (34.6% of the total population) by 2050 [ 8 ]. Similarly, the demographic transition in the Republic of Korea has seen rapid growth in the older adult population. In 2020, 16% of the total population was ≥ 65 years; this is expected to reach 37% by 2050 [ 9 ]. The older adult population made up 13% of the total population in Singapore in 2020 [ 5 ]. The proportion is projected to rise on the same trajectory as China, Japan, and the Republic of Korea in the next few decades [ 1 ].
Improvements in life expectancy have been attributed to better nutrition, political stability, risk factor reduction and improved healthcare access [ 10 ]. Globally, older adults contributed 26% of the total disease burden (measured in disability-adjusted life years [DALYs]); 38% of the burden in high and upper-middle-income, and 16% in low-and lower-middle-income regions in 2019 [ 11 ]. Non-communicable diseases such as cancer, cardiovascular disease and neurological disorders accounted for most of this burden, but respiratory infections, including tuberculosis (TB), are a major contributor as well [ 11 ]. In fact, TB case notifications and estimated disease incidence rates were highest among older adults in China, Japan, the Republic of Korea, and Singapore (Fig. 1 ) [ 12 ].
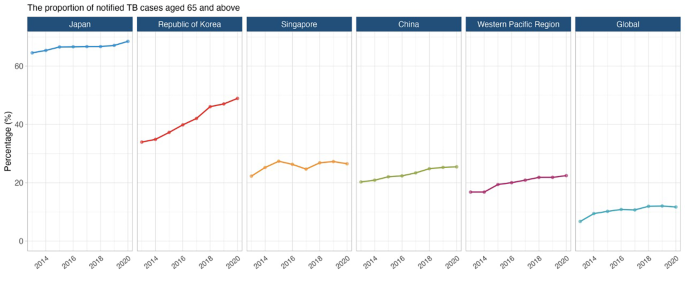
The proportion of notified TB cases aged ≥ 65 in Japan, the Republic of Korea, Singapore, China, the Western Pacific region, and globally 2013−2020
Data source: World Health Organization Global TB Programme 2021
With increasing age, progressive immune dysfunction (immunosenescence) increases the risk for TB disease development [ 13 , 14 ]. The convergence of co-morbidities such as diabetes, chronic respiratory disease and undernutrition, and lifestyle behaviour like tobacco smoking also increase TB risk [ 15 , 16 , 17 , 18 ]. Considering the burgeoning aging population in the Western Pacific Region and the limited guidance specific to this population, it is clear that the needs of older adults require greater recognition in TB response policies. This study reports country case studies from China, Japan, the Republic of Korea, and Singapore, reflecting on country-specific experiences in TB diagnosis and management among older adults. The case studies are a part of a broader endeavour, including a narrative review and analysis of epidemiological trends, to understand and document TB management among older adults in the region. The findings from the narrative review (see supplementary materials for methods) were used to support the country-specific experiences reported in the case studies.
TB epidemiology
China has the second-highest TB burden globally. In 2020, China had an estimated 842,000 people with TB (59 per 100,000 population per year), with older adults accounting for a quarter of all cases [ 12 ]. China notified 159,100 cases (19% of all notified cases) among older adults, [ 12 ] and a consistently increasing trend has been observed over the last two decades [ 19 ]. The fifth national TB epidemiological survey performed in 2010 recorded prevalence rates for clinical and bacteriologically confirmed TB as 482 and 138/100,000 population, respectively, among older adults [ 20 ]. The TB prevalence rate in the survey increased with age and peaked in the 75−79 year age group at 866/100,000 population (overall prevalence rate: 442/100,000 population [95% CI 417−469]) [ 20 ]. A subsequent prospective cohort study reported high TB incidence rates, [ 21 ] highlighting the need to prioritise older adults in the TB response. TB-related mortality was also more prevalent among older adults [ 19 ] concentrated in China’s Central and South-Eastern regions, [ 22 ] among lower-income residents from rural areas and in those with comorbidities [ 23 ].
Health system structure and TB services
The Government of China introduced directly observed treatment (DOT) (now known as treatment support), [ 24 ] as a strategy, in 13 provinces in 1991, with a nationwide scale-up by 2001 [ 25 , 26 ]. The National Center for Disease Control (CDC) is responsible for coordination, policymaking, standards and technical guidance, while provincial CDCs conduct TB programs and monitor disease trends at the prefecture and county level.
TB diagnosis and treatment are provided under the free-TB service policy in China [ 27 , 28 ]. The policy is implemented through an integrated model where designated hospitals (specialised centres for infectious diseases and general hospitals with TB departments) provide TB diagnosis and treatment services [ 29 ]. Persons with drug-resistant TB are treated at designated prefectural or provincial-level hospitals [ 29 ]. Primary healthcare providers are responsible for referring older adults presenting with TB symptoms to designated county-level hospitals for diagnostic investigation. They also conduct follow-up, case management and support, and health education activities at the community level [ 29 ]. The private sector is not involved in TB management. This free-TB service policy is supported by funds from the central and local governments [ 29 ]. Expenditures incurred beyond the free-TB service policy are generally covered by other government-linked insurance schemes [ 27 ]. However, the insurance and financial support schemes vary between provinces, resulting in different levels of financial reimbursement.
Despite this policy, catastrophic costs due to TB remain substantial. Studies report that more than 50% of TB patients incurred out-of-pocket payments exceeding 40% of the household’s ‘capacity to pay, [ 30 , 31 ] indicating a need for better financial support plans and packages to be considered. In addition to financial support, social support through better health education, supportive services, and community engagement is important to improve TB treatment adherence and outcomes [ 32 ].
TB case finding
Active case finding (ACF), a provider-initiated systematic screening and testing for TB disease, [ 33 ] has been deployed in conjunction with the annual physical assessments program for older adults since 2015. Such an integrated approach was feasible and effective for TB case detection among older adults, especially those with comorbidities such as diabetes [ 34 , 35 , 36 ]. TB management is carried out by community health service centres under the technical guidance of the National Tuberculosis Plan [ 37 , 38 ].
Institutionalised aged care is a booming sector in China. As of September 2021, more than 2.6 million older adults were living in retirement facilities [ 39 ]. The quality of services provided in these facilities provides is inconsistent, often resulting in over-crowding in poorly ventilated conditions. Consequently, disease outbreaks are common and in one TB outbreak, 40% of the residents were diagnosed with TB disease [ 40 ]. Therefore, amendments to existing legislation regarding infection prevention and control in such facilities should be considered, with relevant TB information provided to residents and staff. ACF should be prioritised with the expansion of the annual physical assessments program.
TB treatment, care and support have been decentralised using various enablers such as the community, family members, local primary healthcare providers, and technology (electronic medication monitor) for people with TB who were clinically stable and non-infectious [ 26 , 41 ]. However, given their advancing age, existing chronic health conditions, and weakened immunity, older adults are prone to stopping treatment (loss to follow-up) and death [ 42 ]. Currently, limited guidance is provided at the country and global levels to address issues encountered by older adults, such as adverse event monitoring and management, drug-drug interactions and treatment adherence.
Traditional Chinese herbal medicines have been used to treat TB and as adjuvants to improve the tolerability of TB treatment. Although not endorsed by the WHO, herbal medicines are widely used in China [ 43 ]. In 2020, a meta-analysis reported some potential benefits, [ 44 ] but unfortunately, the evaluated studies lacked methodological rigour. Given how widely herbal medicines are used in China, especially in older adults, practice guidelines should consider how best to advise and regulate health practitioners.
TB prevention
The ramp-up of TB response through nationwide expansion of treatment support (historically known as directly observed treatment [DOT]) and improvement of disease reporting and referral system saw a 28% and 65% reduction in the prevalence of pulmonary TB and smear-positive TB, respectively, between 1990 and 2010 in China [ 45 ]. As the risk of infection and transmission decreases, it is expected that reactivation will be the main driver of TB disease occurrence as the population ages in the coming decades [ 46 , 47 ]. China has around 350 million people with TB infection, [ 48 ] however, screening for TB infection and the uptake of TB preventive treatment (TPT) among at-risk groups is limited. Incorporation of TB infection screening into the annual health assessment could improve awareness regarding TB infection and uptake of TPT among at-risk groups but requires further evaluation for feasibility and safety [ 49 ].
The estimated TB incidence in Japan has declined from 36 to 12/100,000 population per year from 2000 to 2020 [ 12 ]. The treatment coverage (notification rate as a proportion of the estimated incidence) was approximately 85%, with 69% reported among older adults (43% among those ≥ 80 years) [ 12 , 50 ]. The median age of a person notified with TB in 2020 was 77 years [ 50 ]. While the number and rate of TB cases have decreased in the last decade, the proportion of older adults among new cases has increased from 48% to 2000 to 59% in 2010 and 69% in 2020 [ 50 ]. Although this is mainly driven by reactivation, multiple outbreaks with transmission in health and aged-care facilities have been reported [ 51 , 52 ]. Despite increased rates of disease, TB mortality among older adults has declined from > 10 to < 5 deaths per 100,000 population in recent years [ 53 ]. Nevertheless, the highest number of TB deaths occurred among older adults in Japan [ 53 ].
The Ministry of Health, Labour, and Welfare of Japan comprises 18 departments, bureaus, and offices that oversee public health, workplace safety and sanitation, medical services, and health insurance [ 54 ]. Access to healthcare in Japan is facilitated by a universal health insurance scheme with population-wide coverage. While approximately 80% of the health facilities are privately owned, they are regulated by the government, and payment for services is controlled by the ministry under the insurance scheme [ 54 ]. These facilities provide direct medical care for people with TB, including in-patient care during TB treatment. Meanwhile, the public health centres provide other aspects of TB response such as screening for TB disease and infection, contact investigation, surveillance, and community-based treatment support [ 55 ]. National TB surveillance data are managed by the Research Institute of Tuberculosis [ 55 ].
TB care is covered by national health insurance and public subsidies at about 70% and 25%, respectively. The remaining 5% is paid out-of-pocket, but a public assistance fund can be used to offset costs for those who cannot afford it [ 55 ]. Medical expenses incurred during hospitalisation are wholly covered by health insurance and public subsidies. Given the rising need for dedicated and specialised care for older adults, a long-term care insurance scheme was introduced in 2020 to support those in need, as well as their family members [ 54 ].
In Japan, TB among older adults has historically been detected passively at outpatient and inpatient health facilities [ 56 , 57 ]. TB case finding among older adults in the health facilities were facilitated by the presence of comorbidities and routine follow-up for other health conditions, thus increasing the likelihood of care-seeking at the onset of TB symptoms and, subsequently, screening for TB [ 58 ]. However, case finding in such settings hinge on a clinician’s awareness of the need to screen for TB. On presentation to health facilities, TB diagnosis among older adults has historically been difficult due to atypical presentation in older adults. For example, one study reported that > 40% of older adults with TB presented with atypical features of TB in Japan [ 59 ]. The non-specific symptoms and low awareness of TB [ 60 ], especially in intermediate TB burden settings such as Japan, could lead to delayed diagnosis.
In addition, community-based ACF programs targeting older adults have been recommended and implemented [ 61 ]. Currently, these programs prioritise older adults ≥ 80 years for TB screening using mobile chest x-rays in the community. For institutionalised older adults, those aged ≥ 65 are offered annual TB disease screening to prevent outbreaks in aged-care facilities. Before admission to an aged-care facility, TB screening using chest radiography has also been implemented to facilitate the early detection of TB disease [ 62 ]. This obligation is also extended to frontline workers in health and aged-care facilities and welfare facilities.
Treatment of drug-susceptible TB in Japan generally follows the standard 6-month regimen [ 55 ] . However, a 9-month regimen without pyrazinamide (2 months of isoniazid, rifampicin, and ethambutol, followed by isoniazid and rifampicin for 7 months) is widely prescribed for older adults, particularly those aged ≥ 80, and the national guidelines did not recommend pyrazinamide for older adults with TB until 2018 [ 63 ]. While recent studies have reported that the pyrazinamide-based regimens do not lead to significantly higher rates of treatment discontinuation, liver injuries, and death than regimens without pyrazinamide, [ 63 , 64 ] the proportion of those receiving the standard 6-month regimen with pyrazinamide declines with age, particularly for those aged ≥ 75 years [ 50 ].
The overall treatment success rate of drug-susceptible TB was 66% in Japan in 2019 [ 50 ] . The treatment success rate among people with TB < 65 years was 82%, and the rate decreased among the older age groups (65−74 years; 76%, 75−84 years; 65%, ≥85 years; 46%) [ 50 ]. The low treatment success rate among older adults has been ascribed to a high death rate during TB treatment. In 2019, 33% of older adults with TB died during treatment, and > 60% died of non-TB-related causes [ 50 ]. Japan has a comprehensive treatment support strategy for people with TB disease or TB infection (TPT). For those who require in-patient care, treatment support that comprises patient education is provided in the hospital. Upon discharge, an individual support plan for community-based treatment support is developed based on the person’s risk of non-adherence, and the frequency and means of medication support are determined. A treatment support conference is held to evaluate and review the plan. Through a patient-centred care approach, treatment support may be provided by health professionals in the local community, community health workers and volunteers, or family members.
Tuberculin skin tests (TST) and interferon-gamma release assays (IGRA) are used to diagnose TB infection in Japan; both tests are covered by national health insurance. TB infection has been a notifiable condition since 2006 [ 50 ]. While there are no policies to actively screen older adults as a priority group for TB infection, notification of TB infection among older adults ≥ 65 has gradually increased since 2010 [ 50 ]. In 2020, approximately 49% of older adults with TB infection were detected via contact investigation, [ 50 ] a national policy to facilitate early detection of TB disease and infection and prevent onward transmission. There was also an increasing number of older adults diagnosed with TB infection in the hospital due to TB infection testing carried out before treating other medical conditions such as rheumatoid arthritis [ 65 ]. Nevertheless, the lower sensitivity of IGRA among older adults remains a concern. A 2017 study in Japan reported discrepancies between IGRA positivity rates and the corresponding estimated prevalence of TB infection among older adults, highlighting the utility and applicability of IGRA in this population [ 66 ]. Furthermore, providing TPT for older adults remains controversial among clinicians due to the risk of adverse events.
- Republic of Korea
In 2020, in the Republic of Korea, there was an estimated TB incidence was 25,000 people with incident TB (49 per 100,000 population per year); 94% of these were notified to the national authorities, of which 49% were aged ≥ 65 [ 12 ]. [ 12 ] While the number of people with TB has steadily declined in the last decade, the proportion of older adults among new TB patients has risen (19.2% in 2001 and 49.1% in 2020) [ 67 ] . In 2020, the TB notification rate among older adults aged 65−69 was 58 per 100,000 population; the rate increased with age and peaked among those aged ≥ 80 years at 235 per 100,000 population [ 67 ]. TB mortality among older adults ≥ 65 was 13.8 per 100,000 population in 2020, the lowest rate recorded since 2001. However, of all TB-related deaths, 82.5% involved those aged ≥ 65, and the proportion has been above 80% since 2016 (2001; 58.0%, 2010; 72.1%). Despite the decline in overall TB burden and mortality rates, the course is predicted to reverse after 2032 (TB incidence) and 2026 (TB deaths) due to increasing trends among older adults, particularly those aged ≥ 80 69 .
The Ministry of Health and Welfare is the national policymaking and governing body for public health and medical services in the Republic of Korea [ 70 ]. The ministry also oversees the national health insurance scheme, a compulsory scheme that confers health care coverage and benefits for the entire population [ 70 ]. The Korean Disease Control and Prevention Agency (KDCA) is responsible for disease surveillance, public health response, disease prevention, and research and oversees the National TB Elimination Project [ 70 ]. While policies regarding TB prevention and care are established by the government, the private sector’s involvement in TB care began in the 1990s and was formalised in 2011 through the public-private mix model [ 71 ].
TB care in the public and private sectors is covered by national health insurance. A 10% co-payment scheme was in place until 2016 [ 71 ]. Since 2017, all expenses incurred during TB care, including hospitalisation, isolation orders (movement restriction to prevent further transmission), and an allowance for dependents, are included in the policy, thereby minimising out-of-pocket payments for people affected by TB. Interventions such as contact investigation and the screening of close contacts for TB disease and infection are also covered by health insurance. While there is specific funding for TB interventions targeted at older adults in the Republic of Korea, the budget only amounted to 1.8% of the total budget for the national TB control program in 2018 [ 71 ].
Since the inception of the national TB control program in the 1960s, early detection and treatment of TB has been a mainstay of TB policies, including for older adults [ 71 ]. In 2020, the TB incidence rate detected through TB screening among older adults ≥ 65 using mobile chest x-ray machines in 17 regions and provinces was 75 per 100,000, 1.9 times higher than the general population’s rate [ 72 ]. This high detection rate prompted KDCA to implement nationwide TB screening among older adults in the community and aged-care facilities as a key strategy to detect TB early in this group.
With an increasingly aging population and the growth in the proportion of women in the labour market, the number of older adults living in institutionalised settings has also increased proportionally, partly due to the introduction of long-term care insurance, which covers home and institutional care services, assistive equipment such as walker and wheelchair, and cash benefits [ 73 ]. In fact, the number of recipients of long-term care insurance increased three-fold between 2008 and 2019 [ 74 ]. Using mobile chest x-ray, TB screening among older adults ≥ 65 in long-term care facilities in 17 regions and provinces resulted in an incidence rate of 66 per 100,000 in 2020 [ 72 ].
Drug-susceptible TB in the Republic of Korea is treated using the standard 6-month regimen [ 75 ]. Partly due to concerns about potential pyrazinamide hepatotoxicity, there is ongoing work to optimise TB treatment among older adults through establishing an adverse events monitoring system, managing adverse events, and developing biomarkers that may predict diagnostic and therapeutic responses [ 76 ].
Overall, the TB treatment success rate has been sitting at approximately > 80% over the last two decades [ 12 ]. However, the treatment success rate among older adults was lower than the general population at approximately 70% between 2012 and 2015 [ 77 ]. Treatment support remains the main treatment administration option. Video-observed therapy and support through a comprehensive support centre for older adults living alone had been trialled with some success. Preliminary data showed higher treatment success rate among older adults enrolled in the programme [ 78 ]. Other incentives have been implemented, such as providing food, daily necessities, and medical accompaniment service for older adults who have trouble remaining on treatment. Furthermore, an approach to assess the vulnerability of people with TB, particularly older adults, with customised case management and linkage with social welfare services have also been implemented.
A comprehensive epidemiologic and contact investigations protocol is in place to screen at-risk populations for both TB disease and infection. TB disease and infection screening is mandatory for healthcare and nursery workers and teachers. Other at-risk groups, such as people living with HIV, those with silicosis, organ transplant recipients, and patients with kidney disease on dialysis, are recommended to be screened for TB infection. For household contacts of persons with TB, chest x-ray investigations and TB infection workups (TST/IGRA, but not mandatory) will be offered (covered by health insurance). TPT (3HP [isoniazid-rifapentine once-weekly for 3 months], 3RH [rifampicin-isoniazid daily for 3 months], or 4R [rifampicin daily for 4 months) is offered to those eligible for it. However, there is no policy to screen older adults for TB infection. In a 2016 study conducted across 11 regions, 40% of those aged ≥60 tested positive for TB infection using IGRA [ 79 ]. Considering the potentially high prevalence of TB infection and risk for reactivation among older adults, [ 80 ] a systematic approach to TB infection screening and TPT administration in this population is warranted.
An estimated 2700 people were affected by TB in 2020 (46 per 100,000 population per year); 89% of these (2400) were notified to the national authorities in 2020. [ 12 ] Of the 2400 people with TB, approximately 57% were Singapore citizens and permanent residents [ 81 ]. The TB incidence rate has been approximately 40 per 100,000 since the late 1990s and may be attributed to migration (about 30−50% of the cases were detected among short and long-term visitors, including work pass holders and students) and an aging population [ 82 , 83 ]. In 2020, older age groups (≥60) made up a significant proportion of the TB notifications both among Singaporean-born (58%) and foreign-born (46%) individuals [ 81 ]. While reactivation of past infections could sustain the TB epidemic among older residents, outbreaks in aged-care facilities have also been reported. Overall, TB mortality rates have remained < 1 per 100,000 population in the past few years [ 12 ]. However, the TB mortality rate among older adults ≥70 was higher at 5.1 per 100,000 population in 2018 [ 84 ].
The Ministry of Health (MOH) Singapore initiated the national TB program, named Singapore TB Elimination Program (STEP), to strengthen TB response efforts by detecting and treating TB disease and infection and preventing drug-resistance TB in 1997 [ 82 ]. STEP manages a notification registry and a treatment surveillance system to monitor TB notification and treatment outcomes. The TB Control Unit (TBCU) is the referral centre for TB management in Singapore, responsible for treatment support, contact investigation, and the administration of TPT. Both public and private hospitals can offer TB diagnosis and treatment. Referrals for TB diagnosis and treatment can be made by primary care physicians in public, private, and community-based organisations (such as the Singapore Anti-Tuberculosis Association).
Singapore’s health system is financed through subsidies, a national health savings account (MediSave), a basic national health insurance scheme (MediShield) for hospitalisation and treatment bills, and an endowment scheme for individuals who have exhausted other means of payment [ 85 ]. For citizens and permanent residents, TB diagnosis and treatment costs are covered by the health financing system. Non-residents, such as those holding long-term work visas, are not covered by these schemes and must rely on employer coverage, private health insurance, or out-of-pocket payment for TB screening and diagnosis costs. TB medications are provided to all at no cost.
TB is diagnosed by passive case finding when a person presents with symptoms or a chest x-ray for other medical conditions (yet suggestive of TB) [ 86 ]. TB screening is conducted for foreigners applying for work or long-term visas and during the visa renewal process using chest radiography [ 86 ]. The same approach applies to everyone regardless of age. There are currently no specific interventions targeting TB screening among older adults. Some aged-care facilities may enquire about the date and information of the last chest x-ray or require a chest x-ray to be conducted before admission. However, this policy is inconsistent.
Treatment of drug-susceptible TB in Singapore follows the standard 6-month regimen (2 months of isoniazid, rifampicin, ethambutol, and pyrazinamide followed by isoniazid and rifampicin for 4 months) [ 86 ]. However, for older adults who may be unlikely to tolerate pyrazinamide, a 9-month regimen comprising ethambutol, rifampicin, and isoniazid for 2 months (intensive phase), followed by rifampicin and isoniazid for 7 months (continuation phase) is used [ 86 ]. It is required by law under the Infectious Diseases Act for all clinicians to report treatment progress and the outcomes of people with TB to the MOH [ 86 ].
The treatment success rate was 79% in 2019, [ 12 ] and the treatment success rate among older adults was not known at the time of writing. Different forms of treatment support have been implemented to support treatment. Outpatient treatment support is implemented at all the 18 public polyclinics and the TBCU. People with TB disease can choose to have their treatment administered at the nearest public health facility. In a 2016 study among adults with TB in Singapore, 72% of respondents reported that they could accept the schedule of facility-based treatment support [ 87 ]. However, 45% perceived the arrangement to be disruptive to their work, school, and social activities [ 87 ]. The results were not age disaggregated. While not specific to older adults, incentive-based intervention to encourage treatment adherence among people with TB from the lower-income bracket has shown promise compared to the non-intervention group in Singapore [ 88 ]. Other forms of treatment support, such as administration in aged-care facilities and through outreach for recipients who have mobility issues or are frail, have been implemented [ 86 ]. Self-administered treatment is also used in Singapore, especially for those treated in the private sector. However, there are no data on self-administered treatment among older adults.
The STEP strategy includes contact investigation to identify close contacts of persons with bacteriologically confirmed TB [ 86 ]. Testing for TB infection is recommended for high-risk groups if there is an intention to treat with TPT [ 86 ]. IGRA is the preferred test for TB infection; variable TPT options are available, including 6 H (isoniazid daily for 6 months), or 4R (daily rifampicin for 4 months) [ 86 ]. Although TB infection has been estimated to affect up to 30% of older adults in Singapore, [ 81 ] there is no policy or specific guideline regarding TPT use for older adults. The risk of adverse events associated with TPT remains a concern, and further studies are needed.
Summary and conclusion
The four case studies presented exemplify the range of practices and challenges in managing TB among older adults in the Western Pacific Region. Here we summarise the key findings and action points based on country experiences:
While passive case finding remains the mainstay intervention contributing to TB case detection in older adults, routine ACF has been implemented in China, Japan, and the Republic of Korea. The optimal use of ACF requires further consideration and assessment in different contexts.
A person-centred approach to TB care was identified as a key theme in all four countries. Nevertheless, limited guidance is available at the country and global levels to address issues older adults encounter (e.g., adverse event monitoring and management, consideration of drug-drug interactions, and treatment adherence). Existing evidence (and evidence gaps) related to the optimal management of older adults with TB require systematic review and guideline development.
There is a lack of standardisation in the approach to TB infection testing and TPT provision. It is important to generate the necessary evidence to inform benefit-risk estimates for managing TB infection among older adults.
The use of traditional medicines is culturally deeply rooted among older adults in this region. Their complementary use in TB care should be explored and carefully considered.
A summary of the approaches implemented by the four countries is outlined in Fig. 2 .
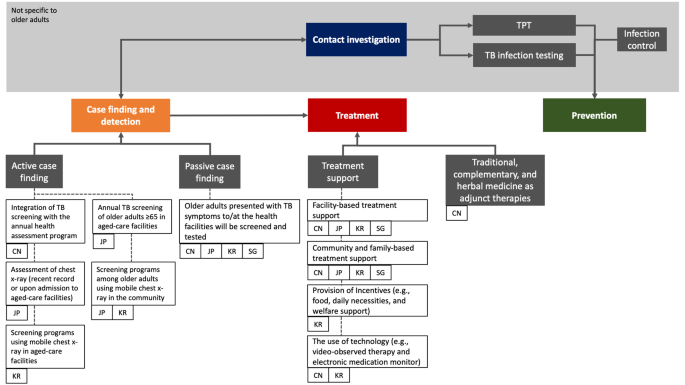
TB management among older adults in China, Japan, the Republic of Korea, and Singapore
TB management experiences were mapped based on three main domains: case finding and detection, treatment, and prevention. The relevant policies and interventions are tagged to the respective implementing countries. Areas shaded in grey represent cross-cutting interventions that extend beyond the older adult population. Treatment support was historically known as directly observed treatment (DOT)
Abbreviations: TB; tuberculosis, TPT; TB preventive treatment, CN; China, JP; Japan, KR; Republic of Korea, SG; Singapore
Cross-cutting challenges
The COVID-19 pandemic has severely disrupted access to TB care. with an 18% decrease in TB disease notifications globally [ 89 ]. Health services for older people were often more severely.
affected and TB services in South-East Asia and the Western Pacific Regions were highly impacted [ 12 ]. Of the four countries included in this study, TB notification data for 2020 and 2021 were lower than 2019 [ 12 , 90 ].
In addition to key risk factors for TB such as living in crowded and poorly ventilated conditions, indoor air pollution and tobacco smoking, [ 91 ] older adults also have age-associated immune dysfunction, financial resources constraints, and frequent comorbidities [ 92 ]. Caring for older adults with TB can be difficult and complex cases may require referral to appropriate specialty care. Service integration between relevant specialties should facilitate better care for older adults with TB. TB diagnosis in older adults is further complicated by challenges related to cognitive impairment, communication challenges and difficulties in obtaining quality sputum samples [ 93 , 94 ]. Dementia, which primarily affects older adults, has been associated with difficult TB diagnosis and poor adherence to TB treatment in the absence of tailored treatment support [ 95 ].
TB-related stigma (perceived and experienced) remains pervasive in many communities [ 96 ]. While systematic assessment of TB-related stigma among older adults has not been conducted, older adults in China have reported unwillingness to seek TB care due to fear of discrimination and self-isolation because of stigma [ 32 , 97 ]. Anecdotally, aged care facilities in Japan were reluctant to accept returning residents after hospital admission for TB care due to fear and stigma, while unwillingness to be screened for TB has also been ascribed to stigma.
The high standards of living and well-being may have shaped the notion that TB is not a significant concern, rendering a lower index of TB suspicion, including among healthcare workers [ 98 ]. A recent cross-sectional study conducted among older adults in Shenzhen, China, reported that > 70% of the respondents were aware of TB as a contagious disease, its symptoms, and preventive measures. Yet, less than half knew that TB was curable, and only one-third were willing to be screened for TB if they were to develop suspicious symptoms [ 99 ].
Considering that aging populations is a global phenomenon that implies a heightened TB risk, particularly in areas with historically high rates of TB, policymakers and funders must invest more to deliver and generate the evidence required to inform optimal TB prevention and care initiatives in older adults.
Data Availability
Data used for this case study comprised published literature, reports, and co-authors’ experience in managing TB among older adults in their respective settings.
Change history
07 june 2023.
A Correction to this paper has been published: https://doi.org/10.1186/s12889-023-15576-0
United Nations Population Division. World Population Prospects. 2019. New York: United Nations Department of Economic and Social Affairs, 2019 (accessed Sept 8, 2021). https://population.un.org/wpp/Graphs/DemographicProfiles/Line/900
World Bank. Population ages 65 and above, total. 2020. (accessed Sept 13, 2021). https://data.worldbank.org/indicator/SP.POP.65UP.TO
The Global Health Observatory. Life expectancy at birth (years). 2020. (accessed Sept 10, 2021). https://www.who.int/data/gho/data/indicators/indicator-details/GHO/life-expectancy-at-birth-(years)
World Bank. Life expectancy at birth, total (years) - Japan. 2020. (accessed Oct 27, 2022). https://data.worldbank.org/indicator/SP.DYN.LE00.IN?locations=JP
World Bank. Population ages 65 and above (% of total population). 2020. (accessed Sept 13, 2021). https://data.worldbank.org/indicator/SP.POP.65UP.TO.ZS?most_recent_value_desc=true
National Institute of Population and Social Security Research. Population projections for Japan (2016–2065). 2017. https://www.ipss.go.jp/pp-zenkoku/e/zenkoku_e2017/pp_zenkoku2017e_gaiyou.html
National Bureau of Statistics of China. The 7th national population census. 2021. (accessed April 1, 2022). http://www.stats.gov.cn/tjsj/tjgb/rkpcgb/qgrkpcgb/202106/t20210628_1818824.html
Department of Economic and Social Affairs, United Nations. World population prospects: population by age and sex (China). 2019. (accessed April 1, 2022). https://population.un.org/wpp/DataQuery/
Korean Statistical Information Service. Projected population by age (Korea). 2021. &tblId=DT_1BPA001&vw_cd=MT_ETITLE&list_id=A41_10&scrId=&language=en&seqNo=&lang_mode=en&obj_var_id=&itm_id=&conn_path=MT_ETITLE&path=%252Feng%252FstatisticsList%252FstatisticsListIndex.do.. https://kosis.kr/statHtml/statHtml.do?orgId=101
Mathers CD, Stevens GA, Boerma T, White RA, Tobias MI. Causes of international increases in older age life expectancy. The Lancet. 2015;385:540–8.
Article Google Scholar
Institute for Health Metrics and Evaluation (IHME). GBD compare data visualizations, Seattle. Washington: IHME, University of Washington, 2021 (accessed Sept 14, 2021). http://vizhub.healthdata.org/gbd-compare
World Health Organization. Global tuberculosis report 2021.Geneva, 2021 https://www.who.int/publications/i/item/9789240037021
Mori T, Leung CC. Tuberculosis in the Global Aging Population. Infect Dis Clin N Am. 2010;24:751–68.
Shaw AC, Joshi S, Greenwood H, Panda A, Lord JM. Aging of the Innate Immune System. Curr Opin Immunol. 2010;22:507–13.
Article CAS PubMed PubMed Central Google Scholar
Uchimura K, Ngamvithayapong-Yanai J, Kawatsu L, et al. Characteristics and treatment outcomes of tuberculosis cases by risk groups, Japan, 2007–2010. Western Pac Surveillance Response Journal: WPSAR. 2013;4:11–8.
Article PubMed PubMed Central Google Scholar
Byrne AL, Marais BJ, Mitnick CD, Lecca L, Marks GB. Tuberculosis and chronic respiratory disease: a systematic review. Int J Infect Dis. 2015;32:138–46.
Article PubMed Google Scholar
Donini LM, ScarDeLLa P, PioMbo L. Malnutrition in elderly: social and economic determinants. J Nutr Health Aging. 2013;17:7.
Bates MN, Khalakdina A, Pai M, Chang L, Lessa F, Smith KR. Risk of Tuberculosis from exposure to Tobacco smoke: a systematic review and Meta-analysis. Arch Intern Med. 2007;167:335–42.
Wang L, Wang W. Temporal Trends in notification and mortality of tuberculosis in China, 2004–2019: a joinpoint and age-period-cohort analysis. Int J Environ Res Public Health. 2021;18:5607.
The Office of the Fifth National TB Epidemiological Survey. The fifth national tuberculosis epidemiological survey 2010. China J Antituberc. 2012;34:485–508.
Google Scholar
Cheng J, Sun Y-N, Zhang C-Y, et al. Incidence and risk factors of tuberculosis among the elderly population in China: a prospective cohort study. Infect Dis Poverty. 2020;9:13.
Amsalu E, Liu M, Li Q, et al. Spatial-temporal analysis of tuberculosis in the geriatric population of China: an analysis based on the bayesian conditional autoregressive model. Arch Gerontol Geriatr. 2019;83:328–37.
Wang L, Xu C, Hu M, et al. Spatio-temporal variation in tuberculosis incidence and risk factors for the disease in a region of unbalanced socio-economic development. BMC Public Health. 2021;21:1817.
World Health Organization. WHO operational handbook on tuberculosis: module 4: treatment: drug-susceptible tuberculosis treatment. Geneva: World Health Organization., 2022 (accessed Jan 25, 2022). https://www.who.int/publications/i/item/9789240048126
Xianyi C, Fengzeng Z, Hongjin D, et al. The DOTS strategy in China: results and lessons after 10 years. Bull World Health Organ. 2002;80:430–6.
PubMed PubMed Central Google Scholar
Hou W-L, Song F-J, Zhang N-X, et al. Implementation and community involvement in DOTS strategy: a systematic review of studies in China [Review article]. Int J Tuberc Lung Dis. 2012;16:1433–40.
Chen S, Zhang H, Pan Y, et al. Are free anti-tuberculosis drugs enough? An empirical study from three cities in China. Infect Dis Poverty. 2015;4:47.
Wei X, Zou G, Walley J, et al. China Tuberculosis Policy at Crucial Crossroads: comparing the practice of different hospital and tuberculosis control collaboration models using Survey Data. PLoS ONE. 2014;9:e90596.
Long Q, Guo L, Jiang W, Huan S, Tang S. Ending tuberculosis in China: health system challenges. The Lancet Public Health. 2021;6:e948–53.
Zhou C, Long Q, Chen J, et al. Factors that determine catastrophic expenditure for tuberculosis care: a patient survey in China. Infect Dis Poverty. 2016;5:6.
Duan W, Zhang W, Wu C, et al. Extent and determinants of catastrophic health expenditure for tuberculosis care in Chongqing municipality, China: a cross-sectional study. BMJ Open. 2019;9:e026638.
Li X, Wang B, Tan D, et al. Effectiveness of comprehensive social support interventions among elderly patients with tuberculosis in communities in China: a community-based trial. J Epidemiol Commun Health. 2018;72:369–75.
Article CAS Google Scholar
World Health Organization. WHO consolidated guidelines on tuberculosis. Module 2: screrening - systematic screening for tuberculosis disease. Geneva: World Health Organization., 2021 (accessed March 9, 2022). https://www.who.int/publications/i/item/9789240001503
Zhang C, Xia L, Rainey JJ, et al. Findings from a pilot project to assess the feasibility of active tuberculosis case finding among seniors in rural Sichuan Province, China, 2017. PLoS ONE. 2019;14:e0214761.
Zhang X-L, Li S-G, Li H-T, et al. Integrating tuberculosis screening into annual health examinations for the rural elderly improves case detection. Int J Tuberculosis Lung Disease: Official J Int Union Against Tuberculosis Lung Disease. 2015;19:787–91.
Li J, Liu X-Q, Jiang S-W, et al. Improving tuberculosis case detection in underdeveloped multi-ethnic regions with high disease burden: a case study of integrated control program in China. Infect Dis Poverty. 2017;6:151.
Office of the State Council. People’s Republic of China. National tuberculosis plan for the 13th five-year plan period. Beijing: Office of the State Council, People’s Republic of China; 2017.
National Health Commission of China. Chinese tuberculosis control and prevention: technical work regulations. Beijing: National Health Commission of China; 2020. http://tb.chinacdc.cn/ggl/202004/P020200414515703939844.pdf
National Ministry of Civil Affairs. Third quarterly report of social welfare institutions 2021. Beijing: National Ministry of Civil Affairs; 2021. http://www.mca.gov.cn/article/sj/tjjb/2021/202103jgqk.html
Hu Y, Hu Y, Ye X. Epidemiological investigation of an outbreak of tuberculosis. Mod Prev Med. 2015;42:982–3.
Wang N, Shewade HD, Thekkur P, et al. Electronic medication monitor for people with tuberculosis: implementation experience from thirty counties in China. PLoS ONE. 2020;15:e0232337.
Abuaku B, Tan H, Li X, Chen M, Huang X. Treatment default and death among tuberculosis patients in Hunan, China. Scand J Infect Dis. 2010;42:281–7.
Tomioka H, Tatano Y, Shimizu T, Sano C. Clinical and basic studies on therapeutic efficacy of Herbal Medicines against Mycobacterial Infections. Med (Basel). 2019;6:E67.
Li X, Li X, Liu Q, et al. Traditional chinese medicine combined with western medicine for the treatment of secondary pulmonary tuberculosis: a PRISMA-compliant meta-analysis. Med (Baltim). 2020;99:e19567.
Wang L, Zhang H, Ruan Y, et al. Tuberculosis prevalence in China, 1990–2010; a longitudinal analysis of national survey data. The Lancet. 2014;383:2057–64.
Huynh GH, Klein DJ, Chin DP, et al. Tuberculosis control strategies to reach the 2035 global targets in China: the role of changing demographics and reactivation disease. BMC Med. 2015;13:88.
Cui X, Gao L, Cao B. Management of latent tuberculosis infection in China: exploring solutions suitable for high-burden countries. Int J Infect Dis. 2020;92:37–40.
Gao L, Bai L, Liu J, et al. Annual risk of tuberculosis infection in rural China: a population-based prospective study. Eur Respir J. 2016;48:168–78.
Article CAS PubMed Google Scholar
Gao L, Zhang H, Xin H, et al. Short-course regimens of rifapentine plus isoniazid to treat latent tuberculosis infection in older chinese patients: a randomised controlled study. Eur Respir J. 2018;52:1801470.
Tuberculosis Surveillance Center. Tuberculosis in Japan: annual report 2021. Tokyo: Department of Epidemiology and Clinical Research, Research Institute of Tuberculosis Japan., 2021 https://jata.or.jp/english/dl/pdf/TB_in_Japan_2021.pdf
Toyota M, Sasaki Y. [The issue of tuberculosis in the elderly in Japan]. Kekkaku. 2010;85:881–94.
PubMed Google Scholar
Seto J, Wada T, Suzuki Y, et al. Mycobacterium tuberculosis Transmission among Elderly Persons, Yamagata Prefecture, Japan, 2009–2015. Emerg Infect Dis. 2017;23:448–55.
Hagiya H, Koyama T, Zamami Y, et al. Trends in incidence and mortality of tuberculosis in Japan: a population-based study, 1997–2016. Epidemiol Infect. 2018;147:e38.
Sakamoto H, Rahman MdM, Nomura S et al. Japan health system review. New Delhi:World Health Organization, Regional Office forSouth-East Asia, 2018 https://apps.who.int/iris/handle/10665/259941
Katsuda N, Hirosawa T, Reyer JA, Hamajima N. Roles of public health centers (hokenjo) in tuberculosis control in Japan. Nagoya J Med Sci. 2015;77:19–28.
Hoshino H, Ohmori M, Uchimura K, Yamauchi Y. [TB epidemiology in an elderly group in Japan]. Kekkaku. 2008;83:423–9.
Kawatsu L, Yoshiyama T, Kato S. Tuberculosis in the Elderly. In: Migliori GB, Raviglione MC, editors. Essential tuberculosis. Cham: Springer International Publishing; 2021. pp. 253–8.
Chapter Google Scholar
Yoshikawa R, Kawatsu L, Uchimura K, Ohkado A. Delay in health-care-seeking treatment among tuberculosis patients in Japan: what are the implications for control in the era of universal health coverage? Western Pac Surveill Response J. 2020;11:37–47.
Hikone M, Ainoda Y, Sakamoto N, Ohnishi K. Clinical characteristics of elderly pulmonary tuberculosis in an acute-care general hospital in Tokyo, Japan: a 12-year retrospective study. J Infect Chemother. 2020;26:245–50.
Nakao M, Sone K, Kagawa Y, et al. Diagnostic delay of pulmonary tuberculosis in patients with acute respiratory distress syndrome associated with aspiration pneumonia: two case reports and a mini-review from Japan. Exp Ther Med. 2016;12:835–9.
Ghotbi N, Nishimura S, Takatsuka N. Japan’s national tuberculosis control strategies with economic considerations. Environ Health Prev Med. 2005;10:213–8.
Ohmori M, Wada M, Mitarai S, et al. [Tuberculosis control in health care facilities for the elderly, from the viewpoint of risk management]. Kekkaku: [Tuberculosis]. 2006;81:71–7.
Hagiwara E, Suido Y, Asaoka M, et al. Safety of pyrazinamide-including regimen in late elderly patients with pulmonary tuberculosis: a prospective randomized open-label study. J Infect Chemother. 2019;25:1026–30.
Takaku T, Saito T, Nemoto K, Oisihi S, Hayashihara K. Comparison of adverse effects in tuberculosis patients over 80 years of age with and without pyrazinamide treatment. Eur Respir J. 2017;50. https://doi.org/10.1183/1393003.congress-2017.PA3045
Kawatsu L, Uchimura K, Ohkado A. Trend and treatment outcomes of latent tuberculosis infection among migrant persons in Japan: retrospective analysis of Japan tuberculosis surveillance data. BMC Infect Dis. 2021;21:42.
Kato S, Ota M, Suenaga M, Hirayama T, Yoshiyama T. Estimation of positive rates of interferon-gamma release assay by age group in Japan. Kekkaku. 2017;92:365–70.
Korea Disease Control and Prevention Agency. Annual report on notified tuberculosis in Korea 2020. Seoul: Korea Disease Control and Prevention Agency, 2021.
Hyewon L, Jinsun K, Hye Kyung I, Youmi K. Characteristics and trends in deaths from tuberculosis in the Republic of Korea 2001–2020. Seoul: Division of Tuberculosis Prevention and Control, Korea Disease Control and Prevention Agency, 2021 &bid=0031&list_no=717657&act=view. https://kdca.go.kr/board/board.es?mid=a30501000000
Jang SY, Kim MJ, Cheong H-K, Oh I-H. Estimating disability-adjusted life years due to tuberculosis in Korea through to the Year 2040. Int J Environ Res Public Health. 2020;17:5960.
Sooman K, Tae-jin L, Chang-yup K. Republic of Korea health system review. Manila: World Health Organization Regional Office for the Western Pacific, 2015 (accessed April 9, 2022). https://apps.who.int/iris/handle/10665/208215
Cho KS. Tuberculosis control in the Republic of Korea. Epidemiol Health. 2018;40:e2018036.
HeeAe K, JeeYeon S, HyeKyung I, EunHye S, WeonJung J, YunSoo K. Results of the 2020 elderly tuberculosis screening in Republic of Korea. Seoul: Korea Disease Control and Prevention Agency; 2021. &bid=0031&list_no=713470&act=view. https://www.kdca.go.kr/board/board.es?mid=a30501000000
Chung Yu L, Euisook K. Long-term care in developing countries: ten case studies (Korea). Geneva: World Health Organization and WHO Collaborating Centre for Health and Long-Term Care Policy and Research; 2003. https://apps.who.int/iris/handle/10665/42769
Ga H. Long-term Care System in Korea. Ann Geriatr Med Res. 2020;24:181–6.
Joint Committee for the Revision of Korean Guidelines for Tuberculosis. Korean guidelines for tuberculosis. 4th ed. Seoul: Korea Disease Control and Prevention Agency; 2020. https://tbzero.kdca.go.kr/tbzero/contents.do
Min J. Study on the optimization of pulmonary TB treatment in elderly 2020–2022. Seoul: Catholic University and the Korean National Institute of Health, 2022 https://cris.nih.go.kr/cris/search/detailSearch.do/17218#step8
Son H, Mok J, Lee M, et al. Status and determinants of treatment outcomes among New Tuberculosis Patients in South Korea: a retrospective cohort study. Asia Pac J Public Health. 2021;33:907–13.
Kim HJ. Enhanced video observed therapy (VOT) to the elderly TB patients: case study of Korea [PowerPoint presentation]. 2021.
Hwang H-J, Park J-H, Nam S, et al. Characteristics of a large latent tuberculous infection screening programme using QuantiFERON®-TB gold in Korea. Int J Tuberc Lung Dis. 2018;22:504–9.
Rajagopalan S. Tuberculosis and aging: A Global Health Problem. Clin Infect Dis. 2001;33:1034–9.
Ministry of Health Singapore. Update on tuberculosis situation in Singapore. Singapore: Ministry of Health Singapore., 2021 https://www.moh.gov.sg/news-highlights/details/update-on-tuberculosis-situation-in-singapore-2022
Loh KS, Hsu LY. Tuberculosis in Singapore: past and future. Ann Acad Med Singap. 2019;48:72–4.
Tam G, Lai SW. Is Singapore on track to eliminate tuberculosis by 2030? A policy case study. SAGE Open Medicine. 2019;7. https://doi.org/10.1177/2050312119851331
Communicable Diseases Division, Ministry of Health Singapore. Communicable diseases surveillance Singapore 2018. Singapore: Ministry of Health Singapore, 2019 https://www.moh.gov.sg/docs/librariesprovider5/diseases-updates/communicable-diseases-surveillance-in-singapore-2018210c9a3beaa94db49299c2da53322dce.pdf
Lim J. Sustainable Health Care Financing: the Singapore experience. Global Policy. 2017;8:103–9.
Ministry of Health Singapore. Prevention, diagnosis, and management of tuberculosis: MOH clinical practice guidelines. Singapore: Ministry of Health Singapore., 2016 https://www.moh.gov.sg/hpp/doctors/guidelines/GuidelineDetails/cpgmed_prevention_diagnosis_management_tuberculosis
Lee HK, Teo SSH, Barbier S, Tang SC, Yeo GH, Tan NC. The impact of direct observed therapy on daily living activities, quality of life and socioeconomic burden on patients with tuberculosis in primary care in Singapore. Proceedings of Singapore Healthcare 2016; 25: 235–42.
Chua AP-G, Lim LK-Y, Ng H, Chee CB-E, Wang YT. Outcome of a grocery voucher incentive scheme for low-income tuberculosis patients on directly observed therapy in Singapore. Singap Med J. 2015;56:274–9.
Migliori GB, Thong PM, Alffenaar J-W, et al. Gauging the impact of the COVID-19 pandemic on tuberculosis services: a global study. Eur Respir J. 2021. 58. https://doi.org/10.1183/13993003.01786-2021
Morishita F. COVID-19 impact on TB notification in the Western Pacific Region. 2022.
Lönnroth K, Jaramillo E, Williams BG, Dye C, Raviglione M. Drivers of tuberculosis epidemics: the role of risk factors and social determinants. Soc Sci Med. 2009;68:2240–6.
Negin J, Abimbola S, Marais BJ. Tuberculosis among older adults–time to take notice. Int J Infect diseases: IJID: official publication Int Soc Infect Dis. 2015;32:135–7.
Sharma M, Onozaki I, Nunn P. TB in older people in Asia: why it is important. Int J Tuberc Lung Dis. 2021;25:521–4.
Hong JM, Lee H, Menon NV, Lim CT, Lee LP, Ong CWM. Point-of-care diagnostic tests for tuberculosis disease. Sci Transl Med. 2022. published online April 6. https://doi.org/10.1126/scitranslmed.abj4124
Bea S, Lee H, Kim JH, et al. Adherence and Associated factors of treatment regimen in drug-susceptible tuberculosis patients. Front Pharmacol. 2021;12:625078.
Courtwright A, Turner AN. Tuberculosis and stigmatization: pathways and interventions. Public Health Rep. 2010;125:34–42.
Wang Y, Feng J, Zhang J, et al. Willingness to seek medical care for tuberculosis and associated factors among the elderly population in Shenzhen: a cross-sectional study. BMJ Open. 2021;11:e051291.
Watts K. Is Australia neglecting the local Topography when it comes to Catastrophic costs and ending tuberculosis? Trop Med Infect Disease. 2018;3:126.
Wang Y, Gan Y, Zhang J, et al. Analysis of the current status and associated factors of tuberculosis knowledge, attitudes, and practices among elderly people in Shenzhen: a cross-sectional study. BMC Public Health. 2021;21:1163.
Download references
Acknowledgements
We would like to thank Jeffery Cutter, Jun Yang Tay, and Shera Tan of the Singapore TB Program for verifying the section on Singapore’s experience.
The author(s) received no specific funding for this work.
Author information
Kerri Viney, Ben Marais, Heejin Kim, Seiya Kato, Yuhong Liu, Catherine W.M. Ong and Tauhid Islam are joint senior authors.
Authors and Affiliations
Saw Swee Hock School of Public Health, National University of Singapore and National University Health System, Singapore, Singapore
Alvin Kuo Jing Teo & Siyan Yi
Faculty of Medicine and Health, University of Sydney, Sydney, NSW, Australia
Alvin Kuo Jing Teo & Ben Marais
World Health Organization, Regional Office for the Western Pacific, Manila, Philippines
Kalpeshsinh Rahevar, Fukushi Morishita, Manami Yanagawa, Kyung Hyun Oh & Tauhid Islam
Division of Infectious Diseases, Department of Medicine, National University Hospital, Singapore, Singapore
Catherine W.M. Ong
Research Institute of Tuberculosis, Anti-Tuberculosis Association, Tokyo, Japan
Takashi Yoshiyama, Akihiro Ohkado, Lisa Kawatsu, Norio Yamada, Kazuhiro Uchimura & Seiya Kato
Korean National Tuberculosis Association, Seoul, Republic of Korea
Youngeun Choi & Heejin Kim
Office of International Cooperation, Innovation Alliance on Tuberculosis Diagnosis and Treatment, Beijing, China
KHANA Center for Population Health Research, Phnom Penh, Cambodia
Center for Global Health Research, Public Health Program, Touro University California, Vallejo, CA, USA
Global Tuberculosis Programme, World Health Organization, Geneva, Switzerland
Kerri Viney
The University of Sydney Institute for Infectious Diseases (Sydney ID) and the Centre of Research Excellence in Tuberculosis (TB-CRE), Sydney, NSW, Australia
Beijing Chest Hospital, Capital Medical University, Beijing, China
Infectious Diseases Translational Research Programme, Department of Medicine, National University of Singapore, Singapore, Singapore
Institute of Health Innovation and Technology (iHealthtech), National University of Singapore, Singapore, Singapore
Division of Infectious Diseases, Department of Medicine, Woodlands Health, Singapore, Singapore
You can also search for this author in PubMed Google Scholar
Contributions
KR, FM, TI, and SY conceptualised the study. AA, TY, AO, LK, NY, KU, YC, ZC, SK, YL, and CWMO contributed to the country experiences reported in this study. AKJT collated the information. AKJT, BM, KV, KR, FM, MY, and KHO wrote the initial draft. All authors critically reviewed the manuscript and approved the final version.
Corresponding author
Correspondence to Kalpeshsinh Rahevar .

Ethics declarations
Ethics approval and consent to participate.
Not applicable.
Consent for publication
Competing interests.
The authors declare that they have no competing interests. The authors alone are responsible for the views expressed in this article, and they do not necessarily represent the views, decisions, or policies of the institutions with which they are affiliated.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article has been updated to correct a typo and a missing disclaimer.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Teo, A., Rahevar, K., Morishita, F. et al. Tuberculosis in older adults: case studies from four countries with rapidly ageing populations in the western pacific region. BMC Public Health 23 , 370 (2023). https://doi.org/10.1186/s12889-023-15197-7
Download citation
Received : 28 October 2022
Accepted : 02 February 2023
Published : 21 February 2023
DOI : https://doi.org/10.1186/s12889-023-15197-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Best practices
BMC Public Health
ISSN: 1471-2458
- General enquiries: [email protected]
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- Trop Med Infect Dis

Active Case Finding for Tuberculosis in India: A Syntheses of Activities and Outcomes Reported by the National Tuberculosis Elimination Programme
Sharath burugina nagaraja.
1 Department of Community Medicine, Post Graduate Institute of Medical Sciences and Research, Employees State Insurance Corporation Medical College, Bengaluru 560010, India
Pruthu Thekkur
2 The Union, South East Asia Office, New Delhi 110016, India; gro.noinueht@htanirss (S.S.); gro.noinueht@iligask (K.D.S.); [email protected] (J.T.); [email protected] (K.S.S.)
Srinath Satyanarayana
Prathap tharyan.
3 BV Moses Centre for Evidence-Informed Health Care, Clinical Epidemiological Unit, Christian Medical College, Vellore 632002, India; moc.liamg@nayrahtpahtarp
Karuna D. Sagili
Jamhoih tonsing.
4 The Global Fund, 1218 Geneva, Switzerland
Raghuram Rao
5 Central TB Division, Ministry of Health and Family Welfare, New Delhi 110001, India; gro.pctnr@roar
Kuldeep Singh Sachdeva
Associated data.
The data used for this synthesis were from two sources: (1) from the state TB programs and partners in a specific data format shared and (2) from the publicly available annual reports of the National TB Elimination Program. The annual reports of the program are available on the website of the National TB Elimination Program. The data from the states and partners are available from the reviewers upon request.
India launched a national community-based active TB case finding (ACF) campaign in 2017 as part of the strategic plan of the National Tuberculosis Elimination Programme (NTEP). This review evaluated the outcomes for the components of the ACF campaign against the NTEP’s minimum indicators and elicited the challenges faced in implementation. We supplemented data from completed pretested data proformas returned by ACF programme managers from nine states and two union territories (for 2017–2019) and five implementing partner agencies (2013–2020), with summary national data on the state-wise ACF outcomes for 2018–2020 published in annual reports by the NTEP. The data revealed variations in the strategies used to map and screen vulnerable populations and the diagnostic algorithms used across the states and union territories. National data were unavailable to assess whether the NTEP indicators for the minimum proportions identified with presumptive TB among those screened (5%), those with presumptive TB undergoing diagnostic tests (>95%), the minimum sputum smear positivity rate (2% to 3%), those with negative sputum smears tested with chest X-rays or CBNAAT (>95%) and those diagnosed through ACF initiated on anti-TB treatment (>95%) were fulfilled. Only 30% (10/33) of the states in 2018, 23% (7/31) in 2019 and 21% (7/34) in 2020 met the NTEP expectation that 5% of those tested through ACF would be diagnosed with TB (all forms). The number needed to screen to diagnose one person with TB (NNS) was not included among the NTEP’s programme indicators. This rough indicator of the efficiency of ACF varied considerably across the states and union territories. The median NNS in 2018 was 2080 (interquartile range or IQR 517–4068). In 2019, the NNS was 2468 (IQR 1050–7924), and in 2020, the NNS was 906 (IQR 108–6550). The data consistently revealed that the states that tested a greater proportion of those screened during ACF and used chest X-rays or CBNAAT (or both) to diagnose TB had a higher diagnostic yield with a lower NNS. Many implementation challenges, related to health systems, healthcare provision and difficulties experienced by patients, were elicited. We suggest a series of strategic interventions addressing the implementation challenges and the six gaps identified in ACF outcomes and the expected indicators that could potentially improve the efficacy and effectiveness of community-based ACF in India.
1. Introduction
Tuberculosis (TB) remains a major public health problem of global concern. In 2019, of the 10 million people estimated to have developed TB worldwide, only 7.2 million people were notified by the National TB Programs (NTP), indicating that around 2.8 million people with TB (28% of the estimated disease burden) were undetected or were detected but not notified [ 1 ]. Detecting TB through passive case finding (PCF) results in diagnostic delays and the suboptimal detection of TB patients in low- and middle-income countries with a high TB burden due to geographic and/or socioeconomic barriers in accessing health facilities [ 2 ].
In its ‘End TB Strategy’, the World Health Organization (WHO) advocated for the ‘systematic screening’ of high-risk population subgroups to increase TB case detection [ 3 ]. ‘Systematic screening’ includes provider-initiated systematic screening for symptoms and signs of TB disease at health facilities or outside health facilities or both [ 4 ]. Systematic screening for TB disease at health facilities is also called ‘intensified case finding’. Systematic screening for TB disease outside health facilities is called ‘enhanced case finding’ or ‘active case finding’, depending on whether the engagement with the target high-risk population occurs at the group or individual level. Educating high-risk groups about TB disease and advising those with symptoms to visit health facilities for diagnosis and treatment is called ‘enhanced case finding’ (ECF). Proactively screening all individuals within high-risk groups outside health facilities for TB symptoms and linking those with symptoms to TB diagnostic services with an intention to diagnose and treat TB cases is termed ‘active case finding’ (ACF).
Many systematic reviews have concluded that systematic screening leads to increased TB case detection compared to PCF [ 5 , 6 , 7 , 8 , 9 , 10 ]. The beneficial effects largely pertain to intensified or targeted case finding among people at high risk for TB in whom the yield of TB cases was high [ 6 , 7 ]. Community-based ACF (or combined ACF and ECF) activities in the general population had uncertain individual and community-level effects, and the benefits of an earlier diagnosis by community-based ACF activities on patient outcomes and transmission were unclear [ 8 ]. However, more recent observational studies and controlled trials in settings with a high TB prevalence indicate that community-based ACF activities could reduce the diagnostic delay, limit out-of-pocket expenditure and reduce TB incidence [ 11 , 12 , 13 ].
Since the benefits of community-based ACF depend on a number of factors, any decisions on ACF components should be based on evidence of an acceptable yield of microbiologically confirmed TB, using diagnostic algorithms to increase the case finding efficiency by considering the expected prevalence, estimated diagnostic test accuracy and the resource availability for specific settings [ 6 , 10 , 14 ].
Active Case Finding (ACF) Campaign under the National Tuberculosis Elimination Programme
ACF activities were initiated in India in 2009 by nongovernmental organisations (NGOs). ACF activities have expanded since 2013. In 2017, the Revised National TB Control Programme (RNTCP) for India emphasized in its “National Strategic Plan for Tuberculosis Elimination—2017–2025”, a series of ACF activities to be implemented in a campaign mode to complement PCF strategies. The key component of ACF in the National Tuberculosis Elimination Programme (NTEP, then renamed RNTCP) involves a nationwide community-based ACF campaign that mobilizes almost the entire general health system to conduct house-to-house screening for TB symptoms in mapped vulnerable target populations for two weeks thrice in a year [ 15 ]. Other components include using expanded definitions of TB symptoms, chest X-rays as a screening test and rapid molecular tests upfront [ 15 ]. A guidance document, updated in 2017, provides detailed information for implementing ACF activities, as well as the targets and quality indicators to monitor the success of the programme [ 16 ].
Table 1 details the expected indicators set by the NTEP for the programme managers of the ACF campaign [ 16 ].
Expected indicators for the active case finding (ACF) campaign in the National Tuberculosis Elimination Programme *.
| Indicator | Expected Proportion |
|---|---|
| Vulnerable population to be mapped per 1 million population | 11% |
| Number in the mapped target population to be screened | >90% |
| Number with presumptive TB among those screened | 5% |
| Number with presumptive TB patients examined (by smear microscopy, CBNAAT or other investigations) | >95% |
| Number with sputum smear-positive test results | 5% (minimum >2% to 3%) |
| Number of sputum smear-negative TB patients examined by chest X-ray and/or CBNAAT | >90% |
| Number with TB diagnosed among those tested | 5% |
| Number of diagnosed TB patients put on treatment | >95% |
* Adapted from the Central TB Division: Active TB case finding. Guidance document [ 16 ]. CBNAAT = Cartridge based nucleic acid amplification test.
Figure 1 depicts the flow of activities envisaged in the ACF campaign in the NTEP guidance document. Each set of activities provides potential intervention points that could be utilised strategically to facilitate TB case detection.

Screening flow chart for active case finding (ACF) in campaign mode under the National Tuberculosis Elimination Programme (NTEP) with intervention points to facilitate case finding and treatment initiation. ASHA: Accredited social health activist; CBNAAT: Cartridge-based nucleic acid amplification test; CTD: Central TB division. Facilitators : 1 Resources, training, motivation; 2 Vulnerable population per million mapped for screening-11%; 3 Strategic enumeration, health education, community mobili-zation; 4 Setting targets, providing incentives, screening at least 90% of target population; 5 At least 5% presumptive TB patients identified through screening; 6 Facilitating diagnostic testing, ensuring >95% with presumptive TB get tested; 7 Quality control; 8 Increased availability, including mobile units; quality control; 9 Sputum smear-positives expected: 5% (at least 2–3%); 10 Sputum smear-negatives examined by chest X-ray and/or CBNAAT: >90%; 11 Increased availability, quality control; 12 TB diagnosed (all forms) among those tested: at least 5%; 13 Initiated on treatment: >95%.
Considering the effort, money and resources involved in implementing community-based ACF activities, we undertook a systematic review to provide timely information about the potential benefits of ACF in India. We sought this information from two sources: (a) information from state NTEP programme managers and implementing partner organizations and (b) data from published and unpublished studies that provided details of the cascade of screened populations as part of community-based ACF campaigns. The results from the latter source, largely from non-programme ACF activities conducted in India, will be reported in a separate publication. The review protocol was registered in PROSPERO, the international prospective register of systematic reviews, on 26 August 2020 and assigned the registration number: CRD42020199854 [ 17 ].
2. Materials and Methods
We followed the guidance provided in the PRISMA statement [ 18 ] in developing the protocol and reporting this synthesis of ACF activities and outcomes reported by the NTEP.
2.1. Types of Studies
We sought programme data from 2017 to 2020 of ACF activities in India from state NTEP managers and data from 2013 to 2020 from implementing partner agencies about ACF activities supported through the NTEP. The Central TB Division, Ministry of Health, Government of India facilitated this process.
2.2. Participants and ACF Activities
We sent a structured, pretested data proforma ( Supplementary Box S1 ) to the programme managers. We requested details of community-based ACF activities targeting the general population, urban slums, urban non-slums, rural areas, hard-to-reach areas, tribal populations, migrant populations, drug users, household TB contacts and paediatric and elderly populations. Data from ACF programmes solely dealing with TB case finding in institutional groups, such as prisoners, healthcare workers, occupational risk groups and patient groups such as people with diabetes seeking care in hospitals or clinics, were not sought.
2.3. Types of Outcome Measures
The data proforma sought:
- A description of the ACF programmes and the diagnostic algorithms used to detect TB cases;
- The outcomes of ACF activities, including the number (and proportion) of people: (a) screened from the vulnerable target population mapped; (b) identified with presumptive TB; (c) tested for TB at the district medical/microscopy centres; (d) diagnosed with all forms of TB (positivity rate or yield) through sputum microscopy, chest X-ray and GeneXpert (cartridge-based nucleic acid amplification tests or CBNAAT); (e) initiated on anti-TB treatment (treatment initiation rate) and (f) completing treatment (treatment completion rate, loss to follow up and mortality). From this data, we estimated (g) the number needed to screen (NNS), which is the number of individuals who were screened to identify one person diagnosed with TB. We also sought (h) data on the impact of ACF on TB notification.
- The challenges encountered during the process of community-based ACF.
2.4. Search Methods
Two authors (KS and SBN) obtained the contact details of all the organisations and agencies that were supported by the RNTCP/NTEP to implement community-based ACF from January 2013 to December 2020 from the Central TB Division (CTD). We mailed the data proformas in the first week of November 2020 to the programme managers and partner agency contacts. We sent reminders for missing data forms and closed data collection by 30 December 2020. We did not receive data for ACF activities from many NTEP programme managers, since they were involved in the response to the SARS-CoV-2/COVID-19 pandemic. We supplemented the information provided by the state NTEP programme managers with national summary ACF data for all states and union territories from the annual TB reports of the NTEP [ 19 , 20 , 21 , 22 ].
2.5. Data Management and Analysiss
One of us (SBN) extracted and collated data from the returned data proformas. PT and SS independently checked the extracted data, and all authors reviewed and discussed the data. We evaluated the adequacy of reporting in accordance with the TIDieR-PHP reporting guidelines for population health and policy interventions [ 23 ].
Since the data were heterogeneous, we tabulated our results in accordance with the synthesis without meta-analysis (SWiM) guidelines [ 24 ]. We assessed the proportions mapped, screened, identified, tested, diagnosed and treated against the expected proportions set by the NTEP for the ACF programme ( Table 1 ). We derived the NNS (from the numbers screened/number diagnosed with TB) for each year for each state and partner agency [ 25 ]. We used the NTEP indicators as a framework to assess the possible associations between the proportions completing relevant parts of the ACF cascade, TB detection rates and the NNS.
We used the information regarding implementation challenges elicited from the programme managers in the returned data proformas and additional information gathered from discussions with the programme managers and partner agencies. We listed them under the broad themes of challenges in implementing ACF activities related to the health system, healthcare provision and those experienced by patients. For further details about the methods used in data management and analyses, see Supplementary Document S1 .
3.1. Respondents
Programme managers from nine states, two union territories and five partner agencies returned completed proformas from among the 28 states, eight union territories and eight partner agencies approached for ACF data ( Figure 2 ).

Flow diagram, in accordance with the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) statement, for the identification and selection of data on community-based active case finding (ACF) activities supported by the National Tuberculosis Elimination Programme (NTEP) of India.
The five NTEP implementing partner agencies were: International Union Against Tuberculosis and Lung Disease (The Union): Project Axshya, the Indian Council for Medical Research (ICMR): Project TIE-TB, the Karnataka Health Promotion Trust (KHPT): Tuberculosis Health Action Learning (THALI) Project, World Health Partners: THALI Project and World Vision India: Project Axshya. Three other partner agencies approached reported no community-based ACF activities in the stipulated time frame.
Though many states did not return completed data proformas, the partner agencies that responded conducted ACF in many unrepresented states.
3.2. Overall Data Quality and Completeness
Four states (Bihar, Gujarat, Karnataka and Maharashtra) provided us with Excel data sheets (in addition to the data proformas) with district and state-wide results of the various rounds of ACF activities conducted from 2017 to 2019. This permitted a comprehensive assessment of the reporting of their ACF activities. The two union territories provided consolidated data for each round of ACF for 2018 and 2019. The remaining states also provided consolidated data (yearly data for 2017–2019—two states and combined data for 2018 and 2019—one state). Two states provided data only for 2017.
The ICMR provided an interim progress report with district-wide data from the five states involved in a 11-month intervention in tribal populations (TIE-TB). The other implementing partners provided combined data from all the years of their respective projects.
We used the available data provided by the states for three years (2017–2019), since the data for 2020 were incomplete or not available.
For 2020, we used the summary data reported in the India TB annual report [ 22 ]. Since the ACF activities during 2020 may have been disrupted due to the pandemic, we compared the patterns in ACF activities for two pre-pandemic years from the India TB reports for 2018 [ 20 ] and 2019 [ 21 ]. Detailed summary ACF data for 2017 were not available in the corresponding annual report [ 19 ].
Deficiencies in reporting were present in about 50% of the returned data proformas ( Supplementary Table S1 ). These were primarily for numerical data for the vulnerable target populations mapped; the proportions screened who were identified with presumptive TB; the proportions with presumptive TB who were tested for TB and the proportions that underwent sputum tests, chest X-rays or CBNAAT. None of the respondents provided numerical data for sputum smear-negative cases or for false positives.
However, all respondents provided numerical data for those with positive tests (sputum, chest X-ray and CBNAAT) and the numbers diagnosed with TB (all forms). It was, therefore, possible to calculate the diagnostic yield (TB prevalence). The NNS could also be estimated for ACF activities in all the states and union territories.
The numbers initiating treatment were unavailable from the data provided by five states. Only three states and the ICMR project provided data on treatment outcomes. The impact of ACF on TB notification was not provided by the states. Among the implementing partners, this was only available for the ICMR TIE-TB project.
3.3. Details of ACF Activities
3.3.1. frequency and duration of acf activities.
The NTEP advocates three rounds of ACF lasting 15 days each year (45 days per year). States used varying strategies for ACF, from biannual rounds for 15 days (or longer), monthly rounds for a variable number of days, ACF activities throughout the year or for one day in a year. The total duration in the states was fewer than the 45 days per year suggested by the ACF campaign. The recommended duration was exceeded by the implementing partners, since ACF occurred daily for the duration of their projects.
3.3.2. Mapping Vulnerable Populations and Selecting Target Areas
Mapping areas for the ACF activities and selecting the target vulnerable populations to screen was based on the guidance provided by the NTEP [ 16 ] and influenced by local knowledge about geographical and occupational vulnerabilities. The prevalence of TB in the populations mapped did not appear to be a formal part of this exercise, except for one partner agency that estimated a variable prevalence of 200–300 per 100,000 population in their areas of activity. The implementing partners worked exclusively among vulnerable high-risk groups, while ACF in the states covered a wider range of vulnerable populations.
3.3.3. Types of ACF Activities
All ACF activities reportedly followed the NTEP guidance and provided health education prior to interviews of household members. Field staff were not always able to screen all members of the household. Many interviewed only the available members, or the head of the household, about symptoms in others. The frequency and proportion of revisits to screen members missed during the initial visits were unclear. In the ICMR’s TIE-TB project, the district TB officers prepared a monthly schedule that was shared with the local primary health centre (PHC) staff to mobilise people who had been screened and presumed to have TB about the monthly visits of the mobile diagnostic vans.
3.3.4. Personnel Engaged for ACF
Apart from community health workers, multipurpose health workers from the general health system and from rural childcare centres (anganwadi workers), workers from the accredited social health activists (ASHA) programme, school wardens, teachers and NGO volunteers were engaged for ACF under the NTEP. Training for ACF personnel using standard guidelines and algorithms was provided over one day by the district programme managers and supervised by the district TB officer and the medical officer for TB control.
The implementing partners recruited and trained project field staff exclusively for ACF activity. Each partner employed different models in delivering ACF activities. For example, The Union’s Project Axshya worked through sub-recipient partners who engaged community volunteers to conduct ACF, with periodic reviews by district and state NTEP managers and external evaluation by the funding agencies. KHPT’s THALI project used community health workers and community structures to implement ACF and trained 19,272 accredited social health activists (ASHA) in ACF activities
3.3.5. Incentives for ACF Personnel and Support for Activities
In larger states, volunteer ACF personnel were provided incentives ranging from 100 to 500 Indian rupees (INR) per day of ACF activity (around 1.3–6.5 US$); one state also provided the team INR 500 per confirmed case. In some states with difficult terrains, the NTEP staff were also provided incentives. One of the partner agencies paid their project staff INR 10 per household screened, one provided incentives only after treatment initiation was documented and three did not provide incentives.
ACF personnel in most states and partner agencies collected sputum samples and transported them to the diagnostic facilities. Under The Union’s Project Axshya, this activity was incentivized with INR 100 per sample transported; however, to ensure the sputum quality, there were limits on the number of samples transported and the sputum positivity rate. In most states, ACF personnel also provided referral forms, travel support or vouchers to people to get chest X-rays done.
The ICMR TIE-TB project exclusively used mobile TB diagnostic vans equipped with sputum microscopy and digital X-ray facilities that visited remote tribal villages once a month. Most states reported that mobile vans for X-rays and for other ACF activities were available, and in one state, the mobile van also had facilities to perform CBNAAT. It was unclear from the data provided how frequently these vans were available and were utilised in ACF activities.
3.3.6. Diagnostic Algorithms Used
Following the initiation of the ACF campaign in the NTEP in 2017, Xpert MTB/Rif tests were deployed up to the subdistrict level in all the states by 2018. By 2019, all states, barring some districts in some small states, reported using the NETP advocated algorithm for sputum smear microscopy and chest X-ray followed by CBNAAT and the TB case definitions provided in Figure 1 . Some also used variations of the diagnostic algorithms, such as sputum microscopy combined with chest X-ray or X-ray and CBNAAT in parallel, while one state performed only smear microscopy and CBNAAT.
The breakdown of the proportion of TB cases diagnosed using each algorithm was not uniformly available from the data provided by the states.
3.4. ACF Activities and Outcomes from Responding State and Union Territories
Table 2 details the activities and outcomes of the active case finding (ACF) campaigns conducted in the states and union territories in India from the available data provided by the NTEP managers for the years 2017–2019.
Activities and outcomes of the active case finding (ACF) campaigns conducted in the states and union territories in India from the available data provided by the National TB Elimination Programme managers (2017–2019).
| State/ Union Territory | Year | Target Population Mapped | Numbers Screened (%) | TB Tested in Those with Presumptive TB (%) and among Those Screened [%] | TB Diagnostic Tests | TB Diagnosed (%; 95% CI) | NNS | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Sputum Positive (%) | X-ray Abnormal (%) | CBNAAT Positive (%) | Anti-TB Treatment Initiated (%) | |||||||
| Andaman & Nicobar | 2017 | 18,526 | 15,040 (81.1) | 11/11 (100) [0.7] | 11/11 (100) | 1/1 (100) | 10/11 (90.9) | 11 (100; 74.1 to 100) | 1367 | 11 (100) |
| 2018 | 1389 | 46 (3.3) | 31/31 (100) [67.4] | 1/23 (4.4) | 0/13 (0) | 1/5 (20) | 1 (3.2; 0.6 to 16.2) | 46 | 1 (100) | |
| Andhra Pradesh | 2018 to 2019 | 34,220,840 | 465,223 (1.4) | 55,922/55,922 (100) [12.0] | 4736/55,922 (8.5) | NA | NA | 4736 (8.5; 8.2 to 8.7) | 98 | NA |
| Bihar | 2017 | 5,650,354 | 3,033,966 (53.7) | NA | 3130/33,754 (9.3) | Nil | Nil | 3130 (9.3; 9.0 to 9.6) | 969 | NA |
| 2018 | 2,722,279 | 1,453,422 (53.4) | NA | 816/24,482 (3.3) | Nil | Nil | 816 (3.3; 3.1 to 3.6) | 1781 | NA | |
| 2019 | 10,298,046 | 6,141,262 (59.6) | 44,858/329,060 (13.6) [0.7] | 2583/31,955 (8.1) | 921/3974 (23.2) | 559/2046 (27.3) | 3200 (7.1; 6.9 to 7.4) | 1919 | NA | |
| Gujarat | 2017 | 14,747,300 | 4,763,436 (32.3) | 37,899/65,059 (58.3) [0.8] | 1331/37,899 (3.5) | 930/6185 (15.0) | Nil | 2261 (6.0; 5.7 to 6.2) | 2106 | NA |
| 2018 | 29,310,663 | 18,452,680 (63.0) | 60,764/79,723 (76.2) [0.3] | 1922/60,764 (3.2) | 1192/15176 (7.9) | 320/4437 (7.2) | 3562 (5.9; 5.7 to 6.1) | 5180 | 1856 (52.1) | |
| 2019 | 59,397,280 | 37,692,373 (63.5) | 77,680/101,304 (76.6) [0.2] | 1889/71,039 (2.7) | 887/20,269 (4.4) | 311/11,892 (2.6) | 3087 (4.0; 3.8 to 4.1) | 12,210 | 1931 (62.6) | |
| Karnataka | 2017 | 12,489,357 | 12,086,328 (96.8) | 110,910/110,910 (100) [0.9] | 4093/110,910 (3.7) | Nil | Nil | 4093 (3.7; 3.6 to 3.8) | 2952 | NA |
| 2018 | NA | 10,265,692 (NA) | 90,041/99,946 (90.1) [0.9] | 1822/85,408 (2.1) | 1914/15,609 (12.3) | 372/1715 (21.7) | 2957 (2.7; 2.6 to 2.8) | 3472 | NA | |
| 2019 | NA | 43,478,614 (NA) | 260,157/307,519 (84.6) [0.6] | 4205/245,243 (1.7) | 4527/42,077 (10.8) | 1836/5747 (40.0) | 7283 (2.8; 2.4 to 2.7) | 5969 | NA | |
| Ladakh (Leh & Kargil) | 2018 | 35,798 | 25,116 (70.2) | 462/NA (NA) [1.8] | 3/374 (0.8) | 0/148 (0) | 13/462 (2.8) | 13 (2.8; 1.7 to 4.8) | 1932 | 13 (100) |
| 2019 | 8996 | 6199 (68.9) | 462/NA (NA); [7.5] | 12/205 (5.9) | Nil | 1/462 (0.2) | 13 (2.8; 1.7 to 4.8) | 477 | 13 (100) | |
| Maharashtra | 2017 | 10,363,469 | 9,413,295 (90.8) | 43,945/55,381 (79.0) [0.5] | 1357/43,945 (3.1) | 2336/17,663 (15.9) | 225/1698 (13.2) | 2654 (6.0; 5.8 to 6.3) | 3547 | 2410 (90.8) |
| 2018 | 23,479,803 | 21,281,430 (90.6) | 74,634/91,225 (81.5) [0.4] | 1925/74,634 (2.6) | 4078/25,283 (16.1) | 411/5209 (7.9) | 3912 (5.2; 5.1 to 5.4) | 5440 | 3845 (98.3) | |
| 2019 | 95,163,760 | 87,568,441 (92.0) | 192,300/211,850 (90.8) [0.2] | 5815/192,300 (3.0) | 27,009/145,805 (18.5) | 1350/23,570 (5.7) | 11,363 (5.9; 5.8 to 6.0) | 7707 | 11,151 (98.1) | |
| Manipur | 2017 | 46,429 | 31,291 (67.4) | 1827/NA (NA) [5.8] | 37/1827 (2.0) | 0/5 (0) | Nil | 37 (2.0; 1.5 to 2.8) | 846 | NA |
| Mizoram | 2017 | 16,8028 | 86,391 (51.4) | 2378/NA (NA) [2.8] | 14/272 (5.2) | 0/5 (0) | 47/2106 (2.2) | 61 (2.6; 2.0 to 3.9) | 1416 | 61 (100) |
| Tamil Nadu | 2017 to 2019 | 8,781,657 | 4,967,754 (56.6) | 1,972,878/ 3,343,099 (59.0) [39.7] | NA/1,972,878 (NA) | NA/1,136,568 (NA) | 2019 data: 277/5969 (4.6) | 6580 (0.3; 0.3 to 0.4) | 755 | 2017 data: 2468/3304 (74.7) |
| Uttarakhand | 2017 to 2019 | 1,412,700 | 125,516 (8.9) | 10,716/NA (NA) [8.5] | 324/10,716 (3.0) | 68/432 (15.7) | 15/600 (2.5) | 407 (3.8; 3.5 to 4.2) | 308 | NA |
CBNAAT = cartridge-based nucleic acid amplification test, CI = confidence interval, NA = not available and NNS = number needed to screen to diagnose on person with TB. a Uncertain if the denominator is the true number of presumptive TB cases identified after screening.
3.5. ACF Activities Provided by Partner Agencies
Table 3 provides details of the activities and outcomes provided by the agencies that implemented ACF in partnership with the NTEP.
Activities and outcomes of the active case finding (ACF) conducted by implementing partner agencies.
| Years | Target Population Mapped | Numbers Screened from Population Mapped (%) | TB Tested in Those with Presumptive TB (%) and among Those Screened [%] | TB Diagnostic Tests | TB Diagnosed (%; 95% CI) | NNS | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Partner Agency | Sputum Positive (%) | X-ray Abnormal (%) | CBNAAT Positive (%) | Anti-TB Treatment Initiated (%) | ||||||
| The Union Axshya Project | 2013-2015 | NA | 8,120,015 households (NA) | 225,443/541,406 (41.6) [NA] | 21,268/225,443 (9.4) | Nil | Nil | 21,268 (9.4; 9.3 to 9.6) | NA | 20,589 (96.8) |
| (The Global Fund) | 2015-2017 | NA | 9,003,299 households (NA) | 272,836/535,613 (50.9) [NA] | 25,493/272,836 (9.3) | Nil | Nil | 25,493 (9.3; 9.2 to 9.5) | NA | 24,524 (96.2) |
| 2018-2020 | NA | 25,575,009 (NA) | 216,075/292,557 (73.9) [0.9] | 15,550/216,075 (7.2) | 4190/10,136 (41.3) | 784/2166 (36.0) | 21,012 (9.7; 9.6 to 9.9) | 1217 | 18,373 (87.4) | |
| ICMR TIE-TB (The Global Fund) | 2015-2017 | 6,117,597 | 55,707 (0.91) | 49,998/49,998 (100) [89.7] | 2091/49,998 (4.2) | 5,272/45,840 (11.5) | NA | 4286 (8.5; 8.3–8.8) | 13 | 4286 (100) |
| KHPT Project THALI (USAID) | 2017-2019 | NA | NA | 21,171/28,473 (74.3) [NA] | 1578/NA | NA | 30/NA | 2247 (10.6; 10.2 to 11.0) | NA | 2174 (96.8) |
| World Health Partners (USAID) | 2017–2019 | 1,707,990 | 381,761 (22.3%) | 6254/6254 (100) [1.6] | 451/6254 (7.2) | Nil | Nil | 451 (7.2; 6.6 to 7.9) | 847 | 451 (100) |
| 2018-2020 | NA | 18,705 (NA) | 1155/1398 (82.6) [6.1] | 46/279 (16.5) | 156/1155 (13.5) | 13/192 (6.8) | 215 (18.6; 16.5 to 21.0) | 87 | 215 (100) | |
| 2019 | NA | 20,863 (NA) | 501/501 (100) [2.4] | 34/501 (6.8) | Nil | Nil | 34 (6.8; 4.9 to 9.3) | 614 | 34 (100) | |
| 2018-2019 | NA | 1389 (NA) | 19/42 (45.2) [1.3] | 1/19 (5.3) | Nil | Nil | 1 (5.3; 0.9 to 24.6) | 1389 | 1 (100) | |
| World Vision (The Global Fund) | 2015-2017 | 3,535,072 | 1.8 million households (NA) | 71,980/NA (NA) [NA] | NA | NA/71,980 | NA | 34,761 (48.4; 48.0 to 48.7) | NA | 34,761 (100) |
CBNAAT = cartridge-based nucleic acid amplification test, CI = confidence interval, ICMR = Indian Council for Medical Research, KHPT = Karnataka Health Promotion Trust, NA = not available and USAID = United States Agency for International Development.
3.6. ACF Outcomes for All States and Union Territories in India for 2020
Table 4 reproduces summary data on ACF activities provided to the NTEP by all the states and union territories in India for the year 2020 that were published in the annual report of the NTEP [ 22 ].
Summary data from the National Tuberculosis Elimination Programme for the active case finding (ACF) activities in 2020 from the states and union territories (ranked by population size) *.
| State/Union Territory (Estimated Population in Millions) | Vulnerable Target Population Mapped from State Population (%) | Numbers Screened from Mapped Target Population (%) | Numbers with Presumptive TB Tested from Those Screened (%) | TB Diagnosed in Those Tested (%; 95% CI) | Number Needed to Screen (NNS) | |
|---|---|---|---|---|---|---|
| 1 | Uttar Pradesh (223.43) | 44,019,832 (18.9) | 43,255,104 (98.3) | 156,980 (0.4) | 10,121 (6.5; 6.3 to 6.6) | 4274 |
| 2 | Maharashtra (125.74) | 85,791,971 (68.2) | 333,161 (0.4) | 311,650 (93.5) | 12,823 (4.1; 4.0 to 4.2) | 26 |
| 3 | Bihar (124.76) | 884,094 (0.7) | 13,776 (1.6) | 49 (0.4) | 7 (1.3; 7.1 to 26.7) | 1968 |
| 4 | West Bengal (99.91) | 13,608,540 (13.6) | 11,997,372 (88.2) | 232,599 (1.9) | 1810 (0.8; 0.7 to 0.8) | 6628 |
| 5 | Madhya Pradesh (84.36) | 14,668,164 (17.4) | 1,070,951 (7.3) | 44,341 (4.1) | 4912 (11.1; 10.8 to 11.4) | 218 |
| 6 | Tamil Nadu (81.4) | 1,148,451 (1.4) | 281,122 (24.5) | 14,744 (5.2) | 395 (2.7; 2.4 to 3.0) | 711 |
| 7 | Rajasthan (79.92) | 8,090,518 (10.1) | 6,906,255 (85.4) | 43,083 (0.6) | 1067 (2.5; 2.3 to 2.6) | 6473 |
| 8 | Gujarat (69.76) | 65,882,010 (94.4) | 50,847,334 (77.2) | 121,466 (0.2) | 4565 (3.8; 3.7 to 3.9) | 11,138 |
| 9 | Karnataka (68.51) | 15,507,273 (22.6) | 92,436 (0.6%) | 87,505 (94.7) | 2939 (3.4; 3.3 to 3.5) | 31 |
| 10 | Andhra Pradesh (52.54) | 1,335,818 (2.5) | 1,151,885 (86.2) | 51,982 (4.5) | 1685 (3.2; 3.1 to 3.4) | 683 |
| 11 | Odisha (46.32) | 45,292,673 (97.8) | 41,965,511 (92.7) | 222,198 (0.5) | 5116 (2.3; 2.2 to 2.4) | 8202 |
| 12 | Jharkhand (39.48) | 14,854,650 (37.6) | 15,230 (0.1) | 10,731 (70.5) | 1891 (17.6; 16.9 to 18.4) | 8 |
| 13 | Telangana (37.92) | 754,912 (2.0) | 60,632 (8.0) | 4822 (8.0) | 1207 (25.0; 23.8 to 26.3) | 50 |
| 14 | Assam (35.05) | 79,329 (0.2) | 15,243 (19.2) | 2029 (13.3) | 91 (4.5; 3.7 to 5.5) | 167 |
| 15 | Kerala (35.44) | 1,171,034 (3.4) | 37,685 (3.2) | 29,166 (77.4) | 802 (2.8; 2.6 to 2.9) | 47 |
| 16 | Punjab (30.67) | 4,856,533 (15.8) | 4,317,208 (88.9) | 5371 (0.1) | 529 (9.9; 9.1 to 10.7) | 8161 |
| 17 | Chhattisgarh (30.03) | 571,344 (1.9) | 7462 (1.3) | 6436 (86.3) | 170 (2.6; 2.3 to 3.1) | 44 |
| 18 | Haryana (29.44) | 9,889,536 (33.6) | 8,282,557 (83.8) | 30,539 (0.4) | 866 (2.8; 2.7 to 3.0) | 9564 |
| UT1 | Delhi (19.05) | 1716 (0.0) | 985 (57.4) | 256 (26.0) | 30 (11.7; 8.3 to 16.2) | 33 |
| UT2 | Jammu & Kashmir (14.50) | 422,954 (2.9) | 141,814 (33.5) | 15,254 (10.8) | 190 (1.3; 1.1 to 2.4) | 746 |
| 19 | Uttarakhand (11.63) | 1,291,237 (11.1) | 1,785,11 (13.8) | 2953 (1.7) | 100 (3.4; 2.8 to 4.1) | 1785 |
| 20 | Himachal Pradesh (7.5) | 7,485,901 (99.8) | 22,709 (0.3) | 15,852 (69.8) | 595 (3.8: 3.5 to 4.1) | 38 |
| 21 | Tripura (3.96) | 198,624 (5.0) | 98,845 (49.8) | 9084 (9.2) | 109 (1.2; 1.0 to 1.5) | 906 |
| 22 | Meghalaya (3.66) | 1,435,077 (39.2) | 532,359 (37.1) | 1064 (0.2) | 28 (2.6; 1.8 to 3.8) | 19,012 |
| 23 | Manipur (3.12) | 53,336 (1.7) | 32,289 (60.5) | 3802 (11.8) | 52 (1.4; 1.0 to 1.8) | 621 |
| 24 | Nagaland (2.07) | 91,005 (4.4) | 23,272 (25.6) | 1291 (5.5) | 23 (1.8; 1.2 to 2.7) | 1011 |
| 25 | Arunachal Pradesh (1.64) | 56,236 (3.4) | 48,925 (87.0) | 2350 (4.8) | 73 (3.1; 2.5 to 3.9) | 670 |
| 26 | Goa (1.54) | NA | NA | NA | NA | NA |
| UT3 | Puducherry (1.50) | 16,152 (1.1) | 10,886 (67.4) | 109 (1.0) | 5 (4.6; 2.0 to 10.1) | 2177 |
| 27 | Mizoram (1.26) | 1,35,399 (10.7) | 59,883 (44.2) | 293 (0.5) | 8 (2.7; 1.4 to 5.3) | 7485 |
| UT4 | Chandigarh (1.17) | 145,297 (12.4) | 6962 (4.8) | 703 (10.1) | 36 (5.1; 3,7 to 7.0) | 193 |
| UT5 | Dadra & Nagar Haveli; Daman & Diu (0.80) | NA | NA | NA | NA | NA |
| 28 | Sikkim (0.66) | 62,853 (9.6) | 11,034 (17.6) | 149 (1.4) | 4 (2.7; 1.1 to 6.7) | 2759 |
| UT6 | Andaman & Nicobar (0.39) | 389,615 (99.0) | 44,762 (11.5) | 432 (1.0) | 21 (4.9; 3.2 to 7.3) | 2130 |
| UT7 | Ladakh (0.34) | 5952 (1.7) | 5952 (100) | 14 (0.2) | 0 (0) | NA |
| UT8 | Lakshadweep (0.07) | 70,070 (100) | 70,070 (100) | 509 (0.7) | 3 (0.6; 0.2 to 1.7) | 23,356 |
| India (1,377.54) | 340,268,106 (24.7) | 171,940,182 (50.5) | 1,429,806 (0.8) | 52,273 (3.66; 3.63 to 3.69) | Median: 906 (IQR 108 to 6550) |
* Modified from Annexure 6 in the India TB report 2021 [ 22 ]. CI = confidence Interval, IQR = interquartile range and UT = Union territory.
The state-wise breakup of the proportions diagnosed with TB by different diagnostic tests was not reported.
3.7. ACF Outcomes for States and Union Terrritories Compared to the Expected Indicators for ACF Set by the NTEP
We used the numerical data provided in these tables ( Table 2 , Table 3 and Table 4 ) and national ACF data for 2018 and 2019 ( Supplementary Tables S2 and S3 ) to evaluate the screening, diagnostic and treatment activities undertaken by the states and union territories and their implementing partners against the expected indicators envisioned by the NTEP for ACF ( Table 1 ).
3.7.1. Vulnerable Target Population Mapped and Screened
The NTEP expects that 110,000 per million vulnerable population (11%) should be mapped for community-based screening.
The national ACF data for 2020 ( Table 4 ) revealed that, despite the disruption caused by the ongoing SARS-CoV-2/COVID-19 pandemic, nearly 25% of India’s 13,378 million population was mapped for screening. This proportion ranged across the states from <1% to 100% (median 10.7%). In 2020, 53% of the states and union territories ( Table 4 ) and 61% in 2019 ( Supplementary Table S3 ) met or exceeded the NTEPs expected indicators for mapping vulnerable populations.
The NTEP expects that >90% of the mapped target vulnerable population should be screened for symptoms of TB.
In 2020, around 51% (range <1–100%; median 37%) of those mapped across the states were screened ( Table 4 ). Eleven states and union territories screened more than 75% of the target population in 2020 but only in 4/34 (12%) with the available data did this exceed the 90% expectation of the NTEP. For 2018 ( Table S2 ) and 2019 ( Table S3 ), the proportion of states and union territories meeting or exceeding this NTEP indicator was 19% and 16%, respectively.
3.7.2. Proportions Undergoing Diagnostic Tests for TB among Those Screened and in Those with Presumptive TB
The NTEP expects that around 5% of people in the community with TB symptoms will be identified through house-to-house screening.
However, the proportions identified with presumptive TB through symptom screening in the states and union territories is not reported in the annual TB reports ( Table 4 , Tables S2 and S3 ).
From the available data for 2017–2019 from the state programme managers, the proportion identified as having presumptive TB from among those screened ranged from <1% in three states, 5% to 10% in three states and 20% to 67% in two others ( Table 2 ). These proportions varied across the three years of reporting. The proportion of responding states that met or exceeded the NTEP expectation for presumptive TB cases identified through ACF ranged from 14% in 2017, 83% in 2018 to 0% in 2019 ( Table 2 ).
The NTEP has not set minimum indicators for the proportion to be tested for TB among those screened ( Table 1 ).
However, the data are reported annually for the proportions tested from among those screened. From 2018 to 2020, <1% of those screened for symptoms of TB in ACF activities across the states and union territories in India underwent TB diagnostic tests ( Tables S2 and S3 and Table 4 ).
The NTEP expects that >95% of those identified with presumptive TB will be tested for TB.
This information is not reported in the annual TB reports ( Tables S2 and S3 and Table 4 ). The available data from the responding states and union territories revealed that 40% (two out of five) in 2017, 29% (two out of seven) in 201 and 17% (one out of six) in 2019 met or exceeded the NTEP indicators for the proportions with presumptive TB who were tested for TB ( Table 2 ).
3.7.3. Diagnostic Algorithms Used and the Proportions Tested with Sputum Smear Microscopy, Chest X-ray and Xpert MTB/Rif (CBNAAT)
The NTEP expects that 5% (minimum >2% to 3%) of those tested would have sputum smear-positive test results.
Sputum smear positivity rates were not available from the national summary tables for the ACF programme ( Tables S2 and S3 and Table 4 ).
Sputum smear positivity rates exceeded the 2% minimum expected in 8/10 responding states and union territories for all or most of the years reported ( Table 2 ). The higher smear positivity seen in the partner agencies data (5.3–16.5%) reflect their focus on screening populations at a higher risk of undiagnosed TB through ACF activities that occurred throughout the year ( Table 3 ).
The NTEP expects that >90% of sputum smear-negative TB patients will be examined by chest X-ray or CBNAAT or both.
This data is not provided in the annual TB reports ( Tables S2 and S3 and Table 4 ).
Most responding states did not report the number of people with sputum smear-negative results who underwent further testing with chest X-rays or CBNAAT or both ( Table 2 and Table 3 ). Where this could be estimated, the data revealed that only 29% (two out of seven) of the responding states in 2017, 50% (three out of six) in 2018 and 20% (one out of five) in 2019 performed chest X-ray or CBNAAT on >90% those with sputum smear-negative results ( Table 3 ).
3.7.4. Proportions Diagnosed with All Forms of TB (Diagnostic Yield)
The NTEP expects that at least 5% of people undergoing diagnostic tests in ACF programmes would be diagnosed with TB (all forms).
Based on the available data from the states and union territories ( Table 2 ), this expectation was met or exceeded by 57% (four out of seven) in 2017, 33% (two out of six) in 2018 and 60% (three out of five) in 2019. The implementing partners exceeded this target for all years of their activities (5.3–48.4%; Table 3 ). Data from more complete nationwide datasets revealed that this expectation was achieved or exceeded in 30% (10/33) of the states and union territories in 2018 ( Table S2 ), 23% (7/31) in 2019 ( Table S3 ) and 21% (7/34) in 2020 ( Table 4 ).
3.7.5. Proportions Initiating and Completing Anti-TB Treatment
The NTEP expects that >95% of TB patients diagnosed through ACF activities should be initiated on anti-TB treatment.
The proportions started on anti-TB through ACF activities in India from 2017 to 2020 were not available from the ACF annual reports published by the NTEP [ 19 , 20 , 21 , 22 ].
The NTEP programme managers stated that, although all diagnosed patients were notified about the NTEP via Nikshay ( https://nikshay.in , accessed on 21 January 2021), the national web-based TB information management system, they could not retrieve treatment information specific to their ACF patients, as Nikshay does not have a dedicated ACF module. Four states and one union territory provided data from their own records of the numbers initiated on treatment ( Table 2 ), and treatment completion ranged from 89% to 100%. The partner agencies reported that 96–100% were started on anti-TB treatment, but none provided data for the treatment outcomes ( Table 3 ).
3.7.6. The Number Needed to Screen (NNS)
The NTEP performance indicators do not include the NNS.
The NNS varied within the states for each year of ACF activity and between the states in the data from the state programme managers ( Table 2 ), implementation partners ( Table 3 ) and national ACF data for 2018–2020 ( Tables S2 and S3 and Table 4 ).
The data uniformly demonstrated that the higher the proportions that are tested for TB among those screened, and the more accurate the tests used to diagnose TB are, the lower the NNS. For example, in 2020 ( Table 4 ), the state with the lowest NNS (Jharkhand) tested 71% of those screened and diagnosed TB in 17.6% of them, resulting in an NNS of 8. The highest NNS of 23,356 was seen in Lakshadweep in the same year, when 0.7% of those screened were tested, and TB was diagnosed in only 0.3% of those tested. Additionally key in this relationship is the risk of TB in the proportions screened and tested.
3.7.7. The Impact of ACF on TB Notification
The NTEP performance indicators do not include the assessment of TB notifications due to ACF programmes.
Data for the impact of ACF on TB notifications in the states and union territories were not provided in the annual TB reports [ 19 , 20 , 21 , 22 ].
Among the partner agencies, only the ICMR TIE-TB project provided data that assessed the impact of the mobile diagnostic units. Of the 24,043 total TB notifications from all sources from October 2017 to June 2018 from the five states that were covered by the project, 3816 (16%) were notified by the mobile diagnostic vans. This proportion varied across the five states (4–25%; median 18%).
The project also estimated that the mean out-of-pocket expenditure for treatment (travel, consultation, investigations, medicines and ancillary costs such as food) was reduced by 78% for patients serviced by mobile vans (average cost INR 255) compared to if they had availed themselves of services through standard government facilities (average cost INR 1163). The reduction in personal expenditure was even greater when compared to treatment at private facilities (average cost INR 6897; 47% spend more than INR 10,000). The project also demonstrated modest reductions in the time to seek consultations, being diagnosed and starting treatment compared to using standard government facilities or private services.
3.8. Challenges Faced by Implementors in Implementing ACF
In Table 5 , we list the challenges expressed by programme managers in implementing ACF, obtained from the responses in the data proformas returned by the programme managers of the state NTEP programmes and the partner agencies and through discussions with some of them.
Challenges in implementing ACF activities as perceived by implementers.
| Category | Challenges | Description |
|---|---|---|
| Health system challenges leading to pre-diagnostic drop-outs and poor documentation of ACF referrals, TB notifications, treatment outcomes and impact of ACF | Poor access to health facilities | Failure to get tested at health facilities due to the distance and time taken to travel, difficulties in finding transport at convenient times, loss of wages incurred due to travel times. |
| Non-availability of all diagnostic tests at peripheral health institutions | Chest radiography and GeneXpert are often not available at one place, but at different levels of health care provision (secondary and tertiary hospitals). This makes it difficult for people to complete the required tests in a day. | |
| Difficulties in accessing radiography services at secondary hospitals | ACF patients are not considered a priority compared to emergency referrals; shortages in materials, resources and equipment malfunction also contribute. | |
| Poor documentation of ACF referrals for diagnostic tests Lack of a separate ACF module in the data management system | Referral slips given by field staff for diagnostic tests are often misplaced by patients or are not entered in diagnostic facilities as an ACF referral. Nikshay, the online data management tool, does not specifically link TB notifications identified by the ACF programme with treatment outcomes. | |
| Healthcare provision challenges leading to poor ACF screening and diagnostic outcomes | Poor TB awareness among general population | Despite time and effort spent on advocacy, communication and social mobilisation, large segments of the vulnerable population are unaware of the importance of the ACF programme and were unwilling to fully comply with ACF requirements. |
| Obtaining an exact denominator of the population, and the geographical boundaries of areas to be mapped | Difficulty in accurately estimating the number of people residing in geographical areas that are mapped. Figures from the previous census are not dynamic and do not accurately reflect the actual population numbers, or its composition, at the time of ACF activities. In many areas, the geographical boundaries of the areas mapped are not clearly demarcated and often overlapped with adjacent areas. | |
| Difficulties due to mountainous terrains and hard-to access areas | Areas in the country with mountainous terrains (as in Leh and Kargil in Ladakh), or other hard-to-reach areas, make it difficult for ACF teams to screen all of the mapped populations. | |
| Challenges faced by patients and families leading to poor compliance with ACF requirements | Pressure to undergo screening and testing | People identified with presumptive TB often do not feel unwell. Requests to visit designated diagnostic centres are perceived as undue pressure from the health workers, particularly if they are busy and if the travel involves long distances and time away from productive work |
| Non-availability of all family members during screening visits | Not all family members can be present when health workers made home-visits. Available family members may find it difficult to accurately report symptoms in other family members. | |
| Non-availability of investigations | Patients are dissatisfied when tests are unavailable when they visit diagnostic facilities, and they have to make multiple visits to complete their tests. | |
| Out-of-pocket expenditure for diagnostic tests | Diagnostic tests are provided free of cost at government-designated facilities. Testing at private diagnostic facilities is often more convenient, but the expenditure involved is considerably greater. |
4. Discussion
India has the highest TB burden in the world. In 2019, India accounted for the largest proportion of people worldwide diagnosed with TB and drug-resistant TB and the largest proportion of under-reported or undiagnosed TB cases [ 26 ]. Implementing a successful community-based ACF programme in a country with a population of over 1.3 billion people is a herculean task. This is the first paper to evaluate the available data from India’s ACF programme against the performance indicators set by the NTEP for ACF and to use the NNS to assess the efficiency of ACF in the states and union territories of the country.
4.1. The Gaps between the Expected Indicators and Outcomes in India’s ACF Programme
This review identified six gaps in India’s ACF programme where the data for the outcomes fell short of the expected performance indicators.
Strategic gap 1: The deficiencies in the proportions mapped from the vulnerable target populations and those screened among the mapped populations.
Strategic gap 2: The discrepancy in reporting the proportions tested among those screened for TB (which is not among the NTEP’s performance indicators), instead of the proportions tested among those identified with presumptive TB (which is a performance indicator for which national data were unavailable).
Strategic gap 3: The deficiencies in ensuring that over 95% of the people that identified with presumptive TB underwent diagnostic testing.
Strategic gap 4: The lack of data to evaluate whether >90% with negative sputum test results underwent additional diagnostic tests.
Strategic gap 5: The deficits in achieving the NTEP’s minimum expected diagnostic yield of 5% TB cases diagnosed among those tested in the ACF programme.
Strategic gap 6: The lack of data from national reports for the proportions initiating and completing treatment in the ACF programmes (and the resultant lack of data to assess the impact of ACF).
In addition to these gaps, the data in this review demonstrate that if a larger proportion of those screened for TB are tested with accurate diagnostic tests, then the NNS would be lower than it currently is in many state ACF campaigns.
4.2. Implications for Potential Interventions to Improve ACF Outcomes and Efficiency
These gaps identify strategic points where various interventions could improve the effectiveness of ACF campaigns.
4.2.1. Improving the Mapping of Vulnerable Populations and Increasing the Uptake of Screening
The gaps identified in mapping vulnerable target populations at a high risk of TB (Strategic gap 1) indicate the need for more accurate and updated TB prevalence data than what is currently available from national and subnational surveys and prevalence studies [ 27 , 28 , 29 , 30 , 31 ]. The challenges experienced by programme managers in mapping vulnerable populations ( Table 5 ) also indicate the need for updated census data and better delineation of the geographical boundaries to be mapped.
One reason identified by programme managers, and echoed in other enquiries [ 32 ], contributing to the low uptake of screening in some areas is the perception in segments of the public about TB. ACF programmes that occur only periodically will have less opportunities to influence public opinion. They also will identify fewer people with undetected TB.
If ACF activities in India are to scale up from campaign mode, more sustained ACF activities must be considered. One option is to integrate ACF with other surveillance activities [ 26 ]. This was successful in 2000 with the active case search and TB-COVID bidirectional screening that enabled TB notifications in India to increase after the lockdowns were lifted [ 22 ]. Scaling up ACF in India also provides an opportunity to align these activities within the broader perspective of the WHO’s multisectoral accountability framework (MAF-TB) [ 33 ]. This would also address the risk factors and determinants of TB and enable collaboration with agencies and stakeholders working on the other sustainable development goals [ 34 ].
Utilising innovative ways to mobilise and use community networks could also be considered. One such approach is the ‘seed-and-recruit’ approach that has been well-received, was deemed feasible and identified more bacteriologically confirmed cases than one-off ACF activities in the countries that have used this approach [ 35 , 36 , 37 ].
4.2.2. Better Use of Data Management Systems
Integrating the available information in Nikshay, the national TB patient management system that also serves as the national TB surveillance system, with geographic information system (GIS) mapping, could provide better estimates of TB prevalence (Strategic gap 1). This data would better inform the state NTEP managers while planning ACF mapping and screening activities, especially in urban areas [ 26 , 38 ].
A dedicated ACF module is currently unavailable in Nikshay, and this was perceived as an implementation challenge ( Table 5 ). A module in Nikshay to document all ACF activities, including the actual number of people in households that were interviewed for symptoms and how many individuals were not, would further address strategic gap 1. The Nikshay mobile app could be used to enter this data by authorised NTEP field staff, as is done by community health workers in other high-TB burdened countries with successful ACF programmes [ 38 ]. Using the mobile app could also streamline the data captured about ACF activities that currently relies on paper forms in many places.
If ACF activities are linked in Nikshay to the diagnostic investigations performed for each person identified through ACF, it would then be possible to generate data on the sputum samples tested. Sputum smear-positive and smear-negative results could be sent to ACF personnel (even through automated messages) to decide on further tests or to facilitate prompt TB notification and treatment initiation and to assess the risk of false-positive diagnoses resulting from screening (Strategic gaps 3, 4 and 6).
This would also permit ACF personnel and managers to review the proportions that did not undergo further tests, assess the reasons for this and encourage a return for tests, with mobile diagnostic units stationed in convenient locations to facilitate this. This would help to reduce pre-diagnosis dropouts (Strategic gaps 3–5).
Linking details of patients diagnosed with TB by ACF in Nikshay and providing real-time access of this data to ACF personnel would also help reduce post-diagnosis drop-outs and provide data about treatment initiation and completion rates (Strategic gap 6).
This information in the ACF module in Nikshay would also provide granular information at a subdistrict level that could be used to assess the impact of ACF activities on TB notifications and treatment outcomes for patients diagnosed by community-based ACF versus more targeted approaches (Strategic gap 6). This information could also be used to track temporal trends in TB identification from different areas in a district and state that can used in refining mapping and screening activities for future rounds of ACF activities (Strategic gap 1).
The India TB report of ACF activities in 2020 contains a bubble plot of the NNS for each state and each high-risk group that was generated through Nikshay using the data provided by the states [ 22 ]. The available NNS data, if linked specifically to ACF activities, could be used to guide strategic planning and implementation decisions to improve the efficiency of the ACF activities.
Making better use of Nikshay for ACF would a cost-effective intervention that will contribute immensely to reducing the six strategic gaps in the ACF cascade identified in this review.
4.2.3. Moving beyond Screening for TB Symptoms
If the aim of ACF is to diagnose people with undetected TB in the community, house-to-house screening for TB symptoms will be insufficient. Many national prevalence surveys across Asia have shown that 40–70% of people detected to have bacteriologically confirmed TB do not report TB symptoms that meet the screening criteria for presumptive TB. Many were detected only because the entire eligible survey population was screened using chest X-rays [ 39 , 40 , 41 , 42 ]. In addition, relying on symptom screening would also miss a large proportion of people with subclinical TB who are usually diagnosed by chest X-ray abnormalities or with molecular techniques [ 43 , 44 ].
This implies that careful consideration should be given to expanding the number of people screened who are offered diagnostic tests, irrespective of whether they have symptoms that meet the criteria for presumptive TB. Selecting the asymptomatic people who are offered additional tests needs to be guided by operational research [ 45 ]. This will help in addressing strategic gaps 1–3.
4.2.4. Increasing the Diagnostic Yield with ACF
The data in this review does not provide clarity on the diagnostic algorithm that would provide the best yield. The use of mobile diagnostic units with digital X-rays and sputum smear microscopy facilities is a pragmatic alternative with the benefits of getting rapid results, as demonstrated by the TIE-TB project. The WHO recommends the use of computer-aided diagnosis (CAD) for interpreting digital X-rays in screening and triage for TB disease in adults over 15 years of age [ 26 ]. The results of operational research should guide the introduction of CAD technologies into scaled-up ACF activities in India.
Expanding the use of Xpert MTB/RIF in ACF programmes is clearly likely to increase the TB notification rates and the numbers initiating treatment [ 39 , 46 , 47 ]. This expansion is likely to be cost-effective compared to using cheaper tests with lower accuracy [ 48 , 49 ]. This will address strategic gap 5 and also contribute to reducing the NNS.
Screening fewer people but testing more of them with accurate diagnostic tools would increase the diagnostic yield and also reduce the NNS (Strategic gaps 1–5). This strategy should be weighed against the current strategy of setting targets to screen large numbers but testing only a small proportion who meet the symptom criteria [ 26 ].
4.3. Limitations of the Review
Some of the limitations of this review relate to the data available from the states and union territories and the partner agencies. Not all states and partner agencies provided the data requested. Additionally, there were lacunae in the data proformas returned by the states and partner agencies.
We also made some changes to the review process after the protocol of this review was registered that was necessitated by the data available for evaluation ( Supplementary Document S1 ).
The challenges faced in implementing ACF were collated from discussions with the NTEP and partner agency programme managers and the responses provided in the data proformas returned by them. These discussions were limited to the programme managers that we were able to reach and, also, did not necessarily capture the difficulties faced by other ACF personnel. They were also not systematic evaluations using formal qualitative methods. However, they provided valuable insights into some of the strategic gaps identified.
5. Conclusions
This review and synthesis of programme activities and outcomes of the ACF campaign launched by the NTEP identified six broad areas where there are gaps between the expectations of the NTEP and the available outcome data from the states and partners implementing ACF. These gaps provided opportunities to intervene strategically, and this review suggests possible interventions that could be considered to improve the efficacy and effectiveness of ACF.
Acknowledgments
We thank the Central Tuberculosis Division, Ministry of Health and Family Welfare, New Delhi and the State Tuberculosis Offices for their timely support in sharing data and information on active case finding conducted by the states. Our special thanks to the implementing partner agencies (The Union, ICMR-TIE TB Project, Karnataka Health Promotional Trust, World Health Partners and World Vision) for sharing the details of the active case finding activities executed in their projects. We acknowledge Paul Garner, READ-IT Director, Liverpool School of Tropical Medicine for his support during the initial phase of prioritising the research question for systematic review.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/tropicalmed6040206/s1 , Box S1: Data proforma, Document S1: Data management and analysis, Table S1: Summary of the completeness of reporting of the ACF activities, Table S2: Summary data from the NTEP for active case finding (ACF) activities in India (2018) and Table S3: Summary data from the NTEP for active case finding (ACF) activities in India (2019).
Author Contributions
Conceptualisation, S.B.N., P.T. (Pruthu Thekkur), S.S., J.T., R.R., K.S.S. and P.T. (Prathap Tharyan); methodology, S.B.N., P.T. (Pruthu Thekkur), S.S., K.D.S. and P.T. (Prathap Tharyan); data collection and validation, S.B.N., P.T. (Pruthu Thekkur), S.S., K.D.S., K.S.S. and P.T. (Prathap Tharyan); formal analysis, S.B.N., P.T. (Pruthu Thekkur), S.S. and P.T. (Prathap Tharyan); data curation, S.B.N., P.T. (Pruthu Thekkur), S.S., K.D.S. and P.T. (Prathap Tharyan); writing—original draft preparation, S.B.N.; writing—review and editing, P.T. (Prathap Tharyan), P.T. (Pruthu Thekkur), S.S., K.D.S., K.S.S. and S.B.N.; visualisation, P.T. (Prathap Tharyan), P.T. (Pruthu Thekkur), S.S. and S.B.N.; supervision, P.T. (Prathap Tharyan) and S.S.; and project administration, K.D.S. All authors have read and agreed to the published version of the manuscript.
This publication is associated with the Research, Evidence and Development Initiative (READ-It). READ-It (project number 300342-104) is funded by UK aid from the UK government; however, the views expressed do not necessarily reflect the UK government’s official policies.
Institutional Review Board Statement
Not Applicable.
Informed Consent Statement
Data availability statement, conflicts of interest.
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of the data; in the writing of the manuscript or in the decision to publish the results.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

COMMENTS
Introduction. Tuberculosis (TB) is caused by strains of Mycobacterium tuberculosis (M. Tuberculosis). TB is a primarily pulmonary infection spread by airborne droplet transmission. The development and spread of drug-resistant strains of M. tuberculosis greatly jeopardize TB control efforts. In the US, 91 cases of MDR-TB were reported in 2014.
Tuberculosis | Infectious Diseases: A Case Study Approach
Personal Stories from TB Survivors - My Journey fighting TB. 3 June 2020. Tuberculosis (TB) is the world's top infectious killer today. It is airborne and can affect any one of us. Over 5 000 women, men and children still die each day from TB. The social and economic impacts are devastating, including poverty, stigma and discrimination.
case study on pulmonary tubercul osis Vedha Pal Jeyamani.S 1 , Angel.P 1 , Bhavya.E 1 , Balaji.P 1 , Muruga n.M 2 1 Department of Pharmacology , Ja ya College of Pharmacy, Chennai, T amilnadu, India
TB Case Study Example - LSU School of Medicine
The Mycobacterium tuberculosis complex includes the following mycobacteria, which are characterized by a slow growing rate: M. tuberculosis, M. africanum, M. bovis, and M. microti ().In recently published reports of two cases of lymphatic node tuberculosis (TB), the strains were recognized as belonging to a new taxon of M. tuberculosis (2,3).These isolates were characterized by a highly ...
Background Mycobacterium tuberculosis (Mtb) has been found to persist within cavities in patients who have completed their anti-tuberculosis therapy. The clinical implications of Mtb persistence after therapy include recurrence of disease and destructive changes within the lungs. Data on residual changes in patients who completed anti-tuberculosis therapy are scarce. This case highlights the ...
Community-based active case-finding interventions for ...
Clinical and radiological manifestations of tuberculosis (TB) are heterogeneous, and differential diagnosis can include both benign and malignant diseases (e.g., sarcoidosis, metastatic diseases, and lymphoma). Diagnostic dilemmas can delay appropriate therapy, favoring Mycobacterium tuberculosis transmission.We report on a case of TB in an immunocompetent, Somalian 22-year-old boy admitted in ...
Although tuberculosis (TB) is a curable disease, treatment is complex and prolonged, requiring considerable commitment from patients. This study aimed to understand the common perspectives of TB patients across Brazil, Russia, India, China, and South Africa throughout their disease journey, including the emotional, psychological, and practical challenges that patients and their families face.
In this case study, a nurse presents her reflections on the challenges of supporting a patient through his treatment journey for multidrug-resistant tuberculosis. The patient has significant comorbidities and social issues, such as diabetes and homelessness. There was also a language barrier. All these aspects made the management of his ...
Case Study 3 TB Infection with Adverse Drug Reaction.....31 Participants will learn about the persons who are at high risk for exposure to tuberculosis, the current methods for diagnosing latent TB infection, treatment of patients with latent tuberculosis infection, and potential adverse reactions and responses to medications used ...
In The Lancet Public Health, Rachael Burke and colleagues 7 report findings of a systematic review summarising the evidence for population-level effectiveness of active case-finding for tuberculosis. The authors appraised 36 studies published between Jan 1, 1980, and April 13, 2020, of adult populations from 16 countries that were exposed to different active case-finding interventions.
improve adherence. A case study highlights patient-centered TB case management. Chapter 3: Tuberculosis Nurse Case Management in Special Situations and Circumstances This chapter provides information and case studies on TB in selected situations and circumstances, ranging from TB in people experiencing homelessness to those with TB and diabetes.
The 2023 epidemiological review found excellent progress in case finding and reporting, with the absolute number of total case notifications increasing from 103 018 in 2018 to 285 561 in 2022, corresponding to an increase in estimated treatment coverage from 24% in 2018 to 59% in 2022 Fig. 1. Among the 282 184 new and relapse TB cases notified ...
However, during the course of this study, both bedaquiline and linezolid have been increasingly used to treat MDR tuberculosis and XDR tuberculosis, and the WHO recently published guidelines that ...
The Western Pacific Region has one of the fastest-growing populations of older adults (≥ 65 years) globally, among whom tuberculosis (TB) poses a particular concern. This study reports country case studies from China, Japan, the Republic of Korea, and Singapore reflecting on their experiences in managing TB among older adults. Across all four countries, TB case notification and incidence ...
Lesson 3: Testing for TB Infection. HOME. GLOSSARY. EXIT. NEXT. BACK. Centers for Disease Control and Prevention 1600 Clifton Rd. Atlanta, GA 30329, USA ... Page 19 of 23. CASE STUDY. Now that you have learned how people are tested for TB, apply your knowledge with the following case study. ...
National Tuberculosis Center2303 Southeast Military Drive San Antonio, Texas 78223 Phone (800) 839-5864 (1-800-TEX-LUNG) Fax (210) 531-4590 Website: htp://www.HeartlandNTBC.org This collection to f nursing case studies and their accompanying tools are intended to complement a TB program's education and.
CASE STUDY. David's health care provider refers him to the local health department for further evaluation. At the health department, David answers questions about his medical history and current medications. He also receives a chest x-ray, a physical examination, and coughs some sputum into a container.
TB CASE STUDY #1. A 31-year-old caucasian male presented to the Emergency Department (ED) after experiencing gross hemoptysis. He had a 2 month history of productive cough, a 25 pound weight loss, night sweats, and fatigue. A CXR revealed bilateral cavitary infiltrates. The initial sputum specimen was smear positive 4+ and was submitted for a ...
Lesson 6: Treatment of TB Disease. HOME. GLOSSARY. EXIT. NEXT. BACK. Centers for Disease Control and Prevention 1600 Clifton Rd. Atlanta, GA 30329, USA ... Lesson 6 Case Study. Page 20 of 29. CASE STUDY. Now that you have learned about the treatment of TB disease, check your knowledge with the following case study. ...
Active Case Finding for Tuberculosis in India